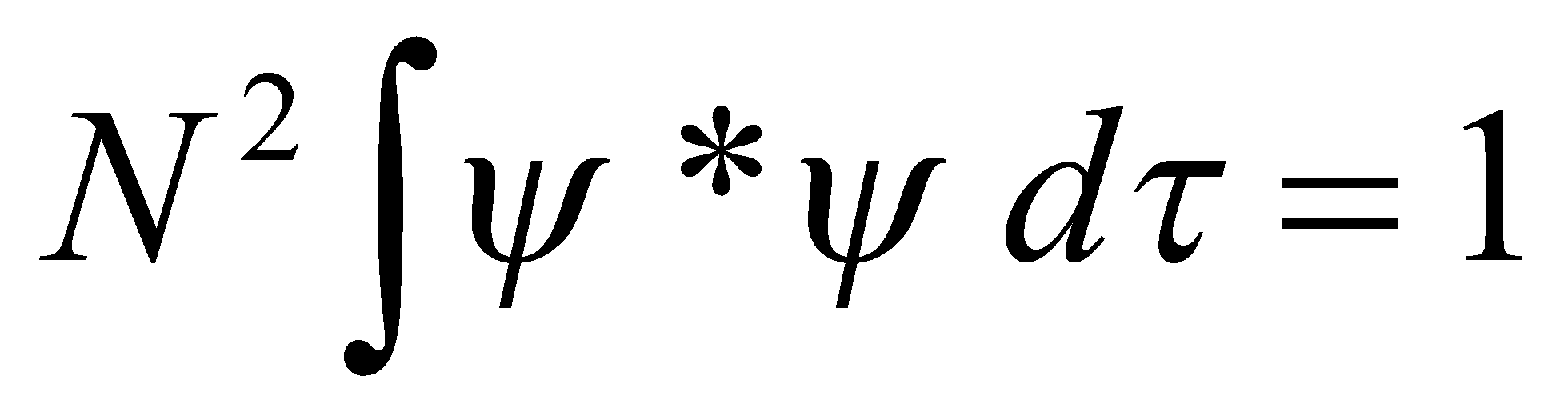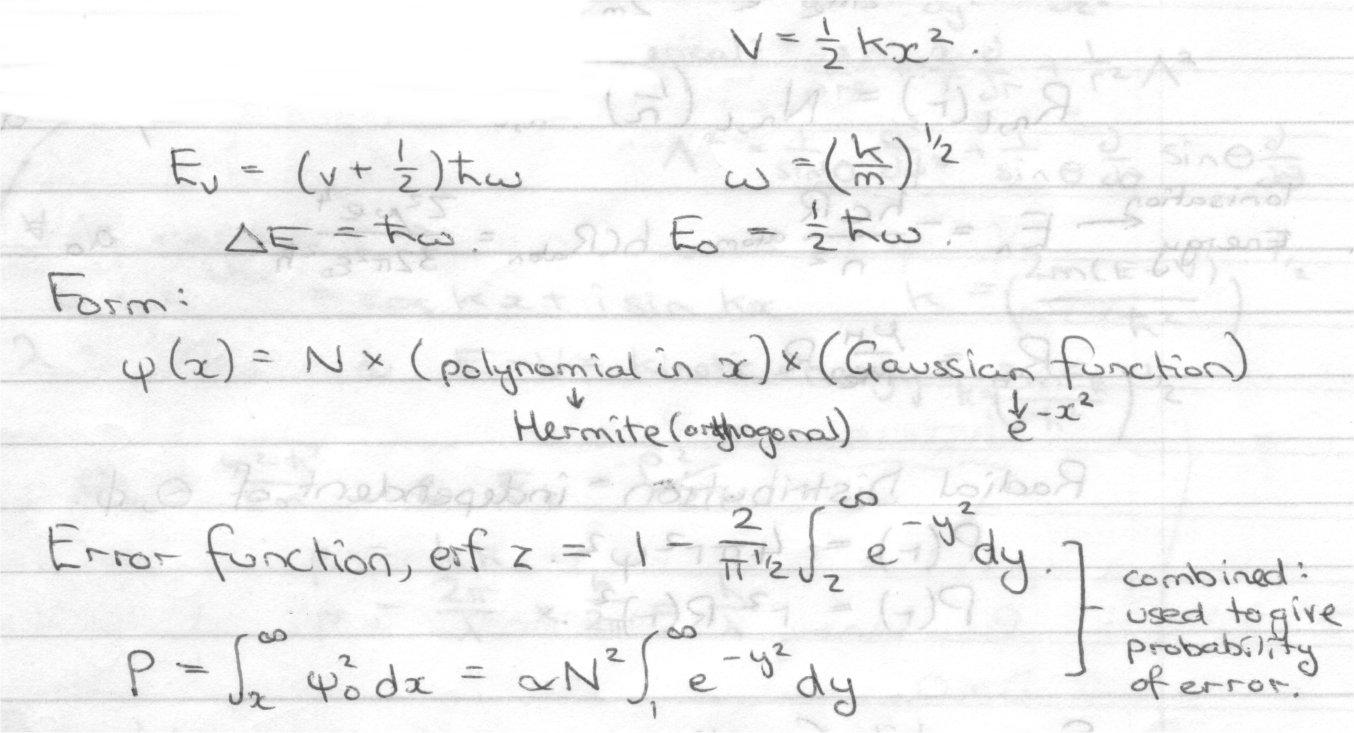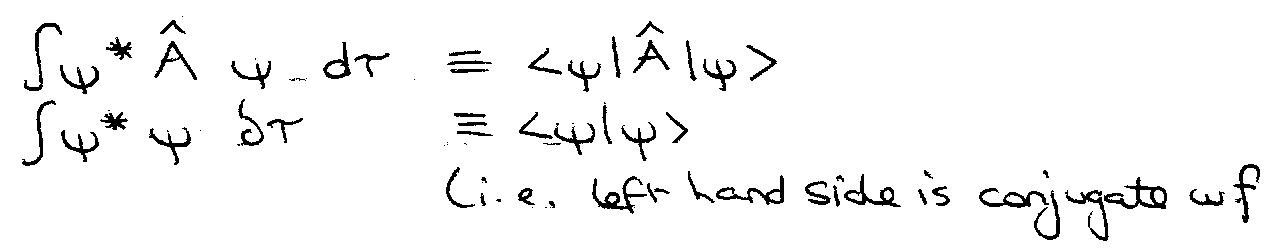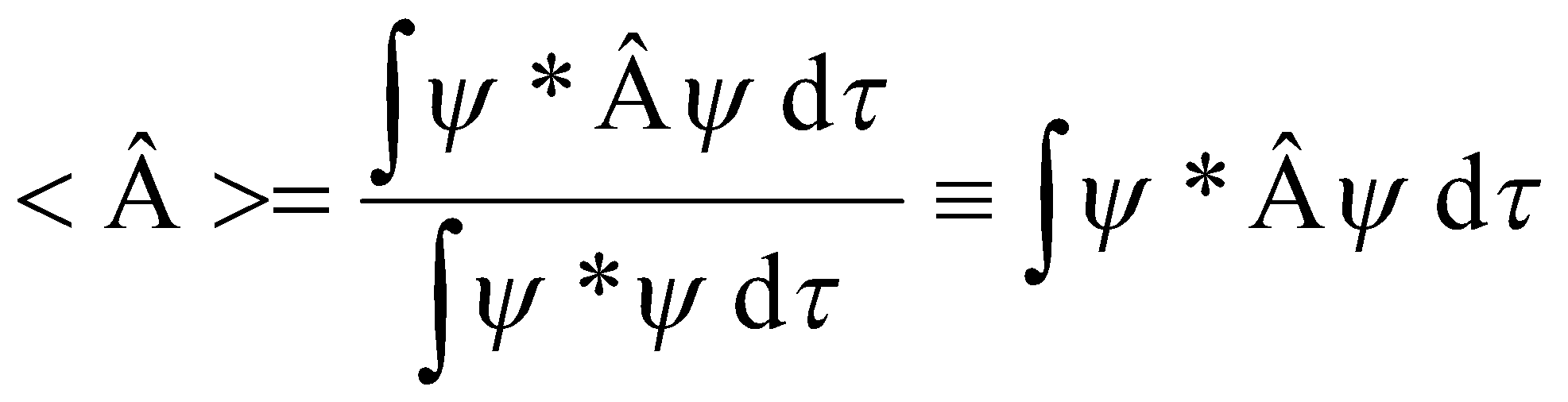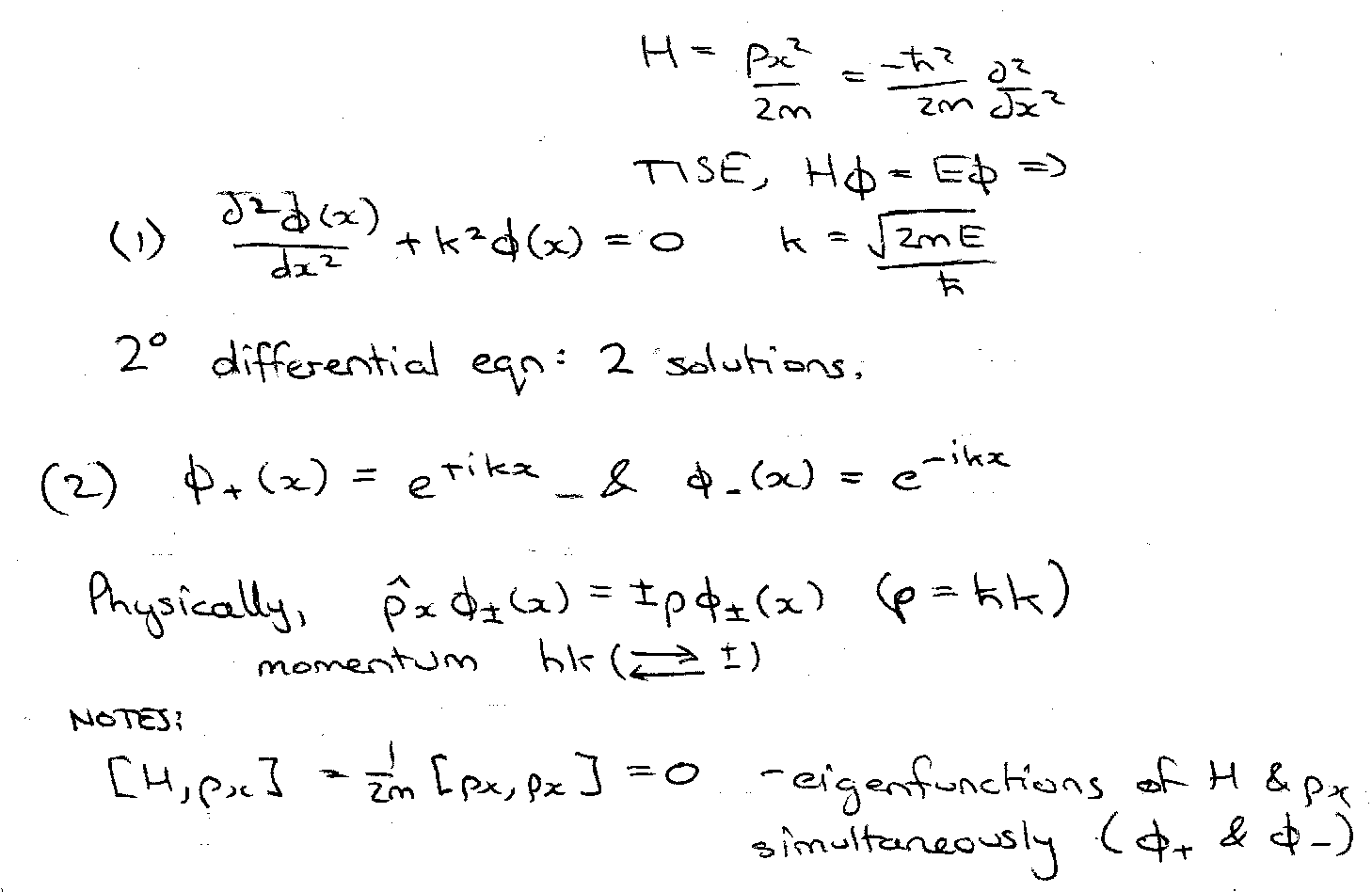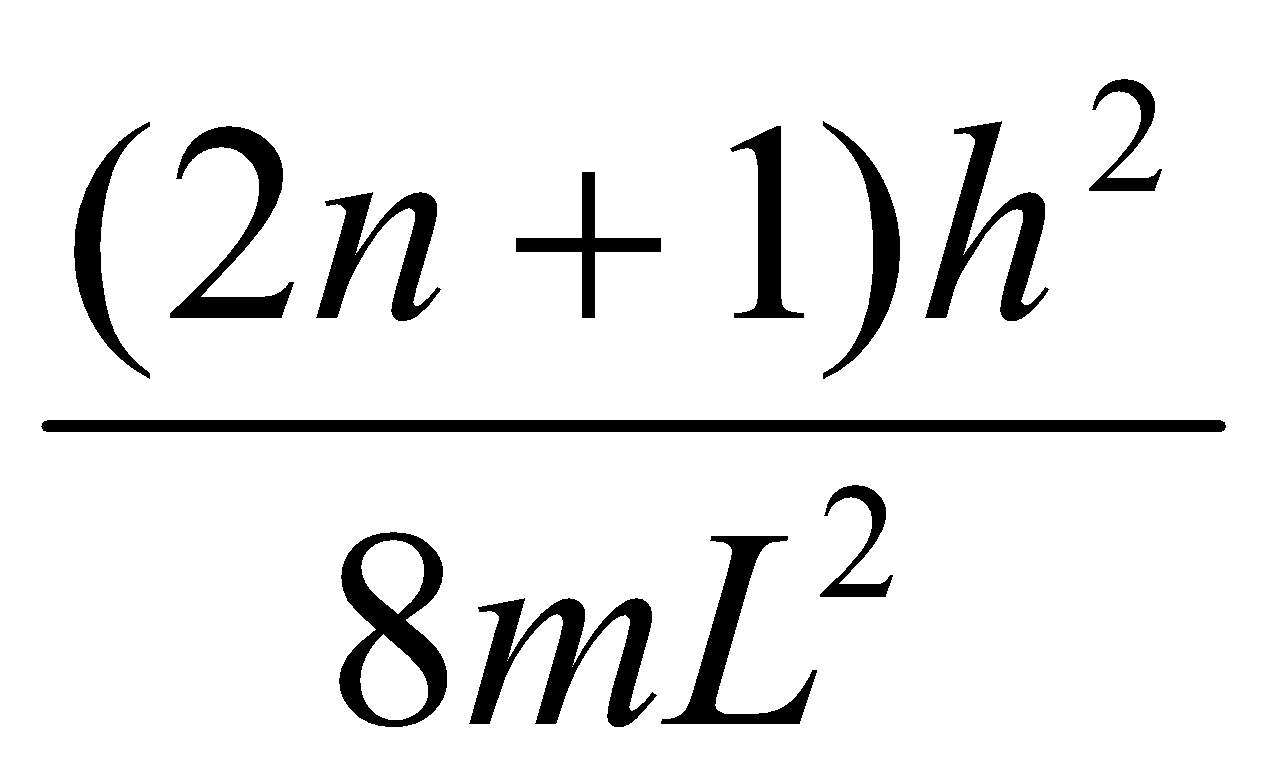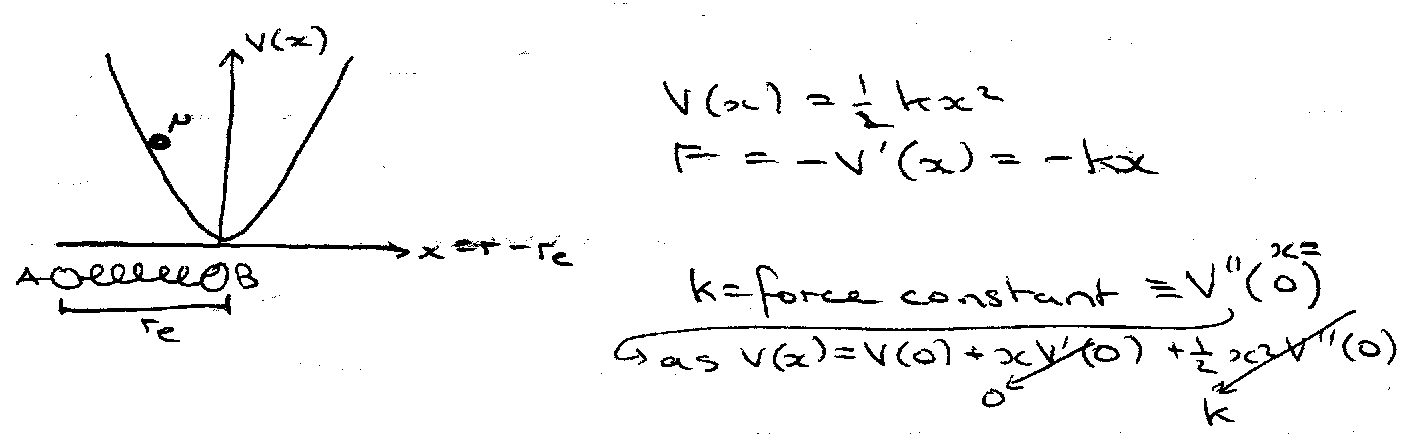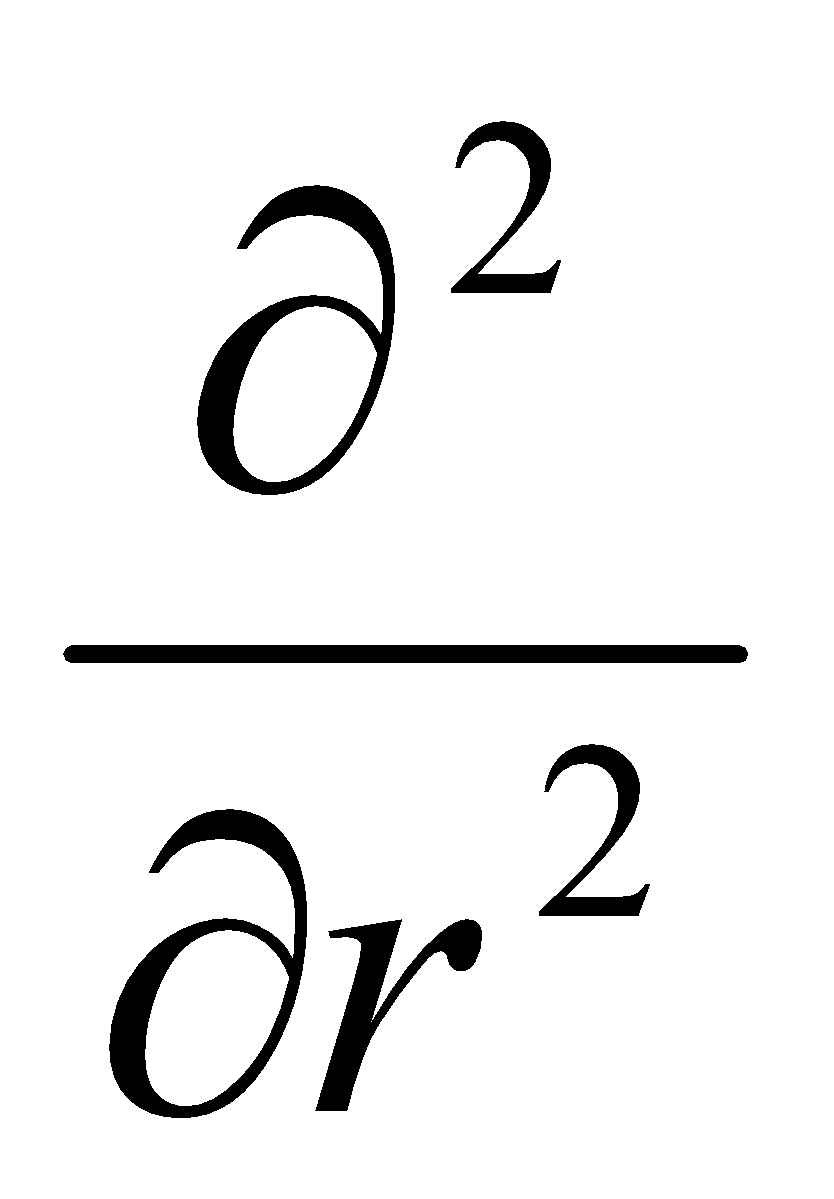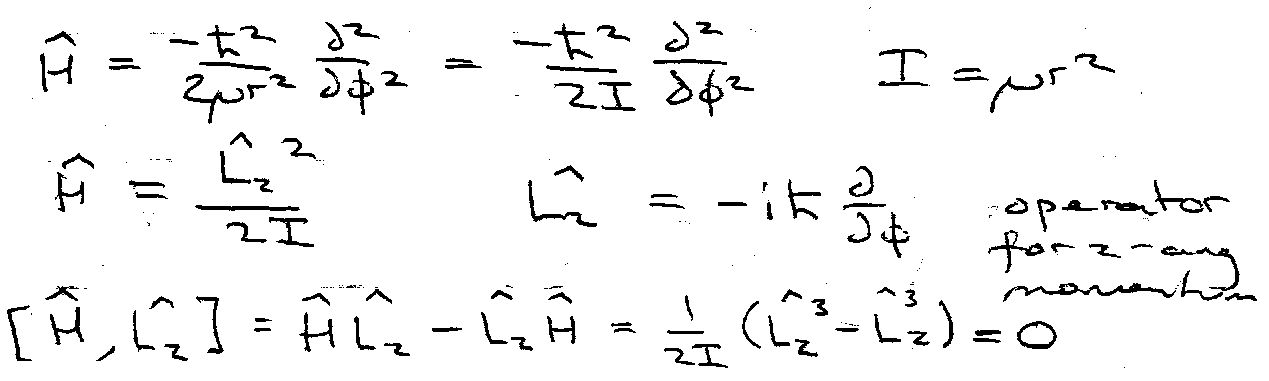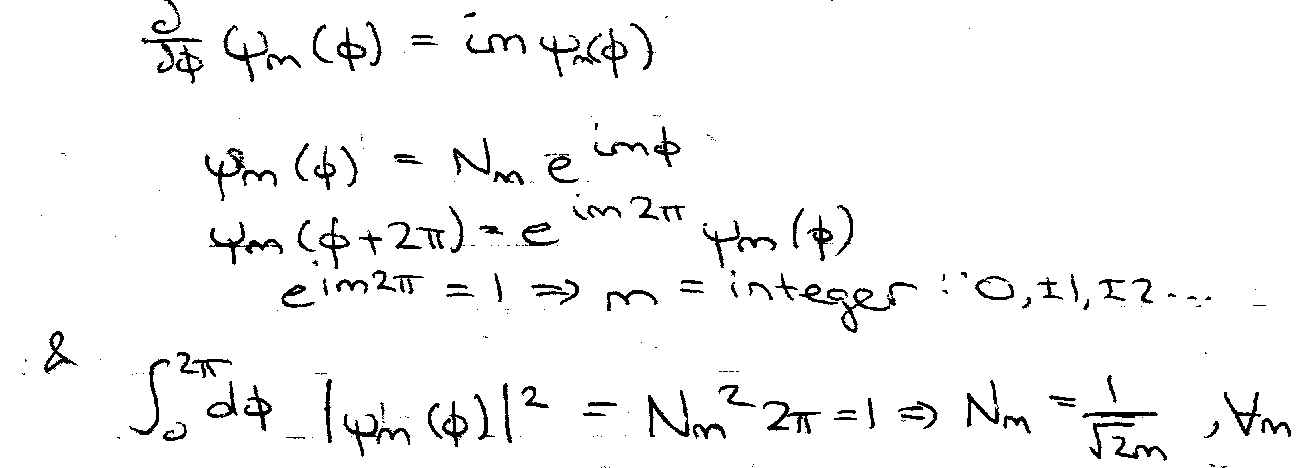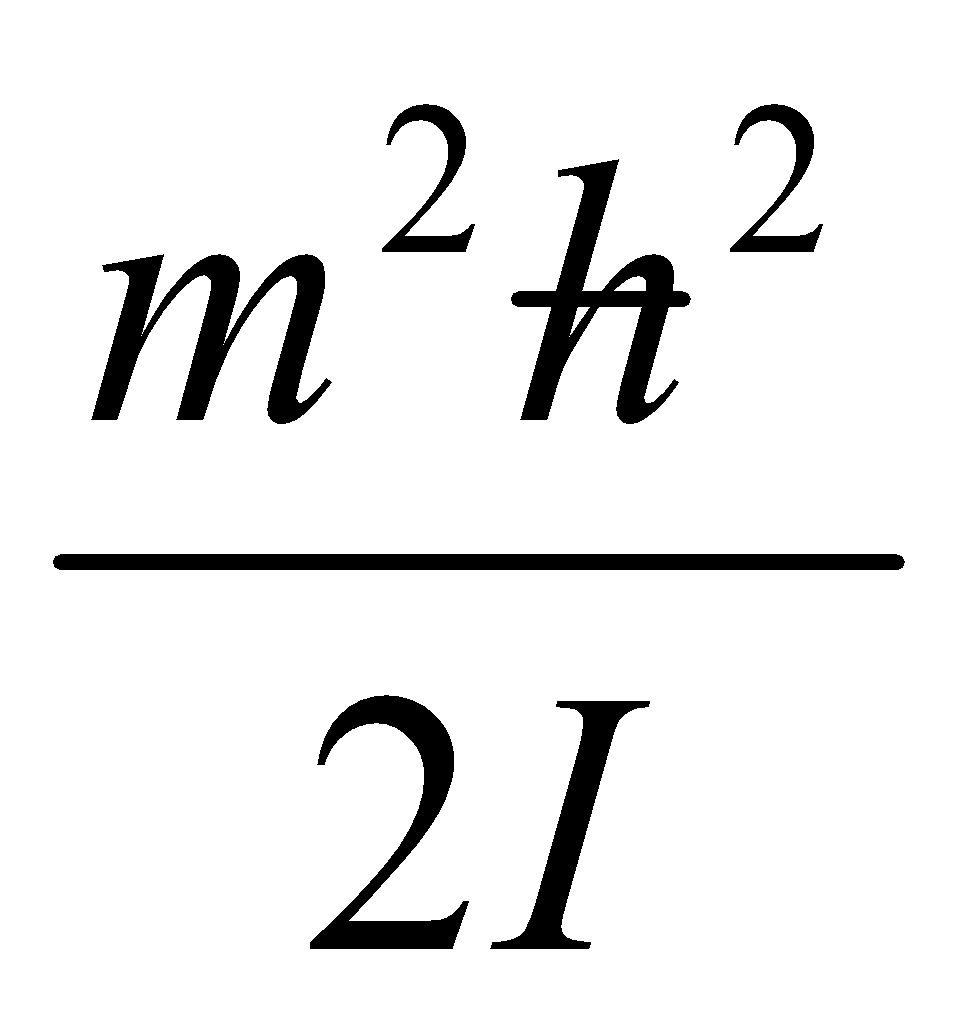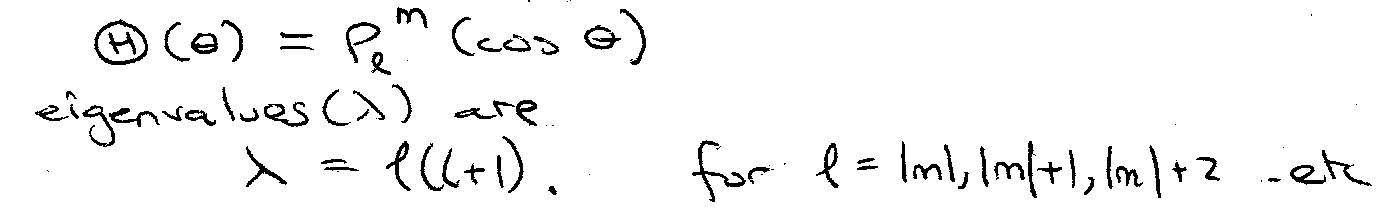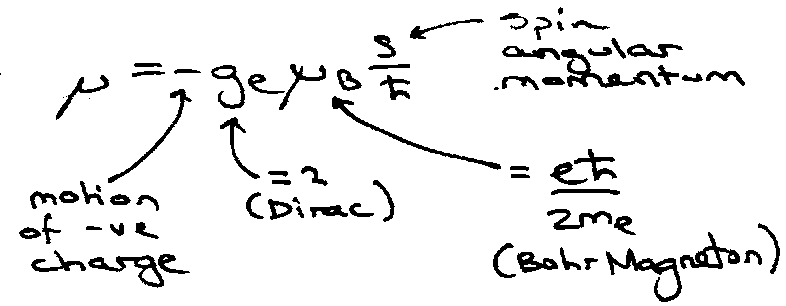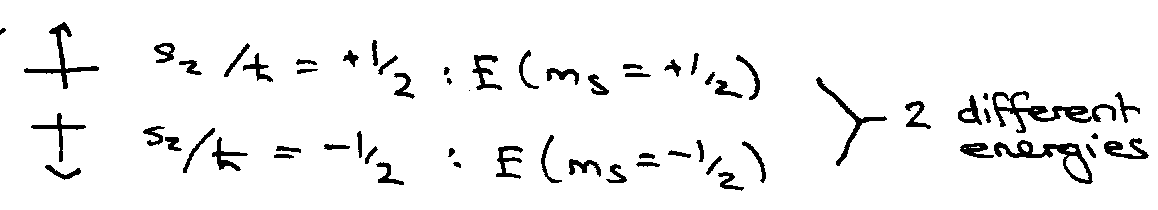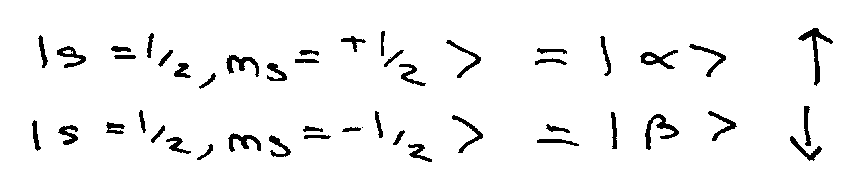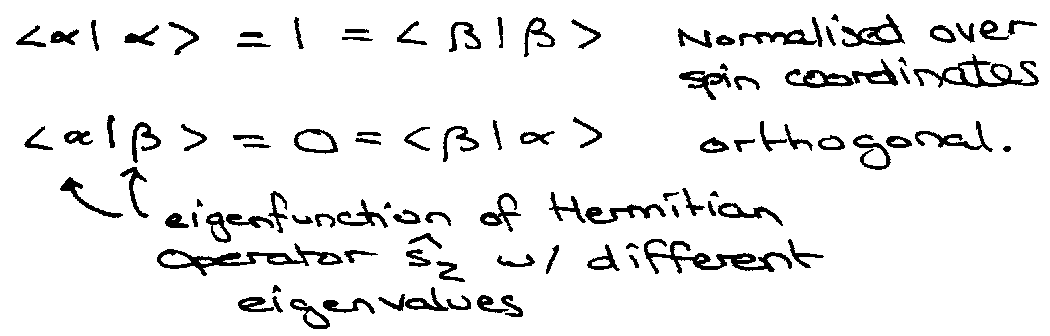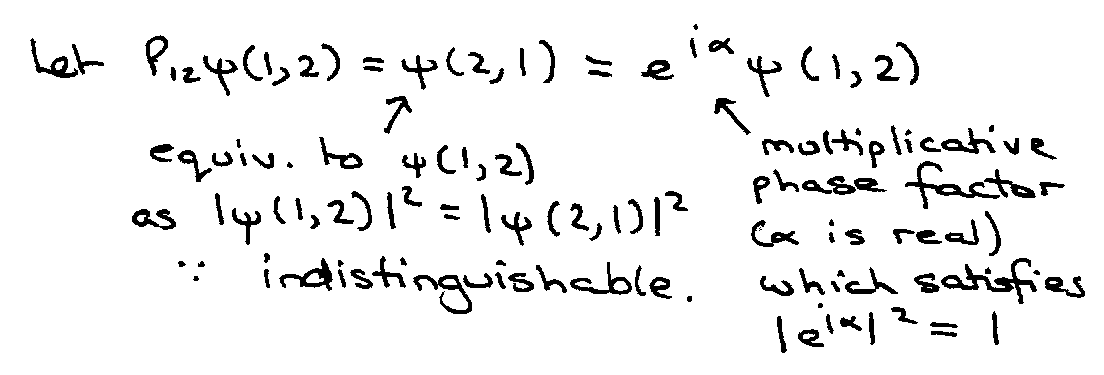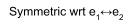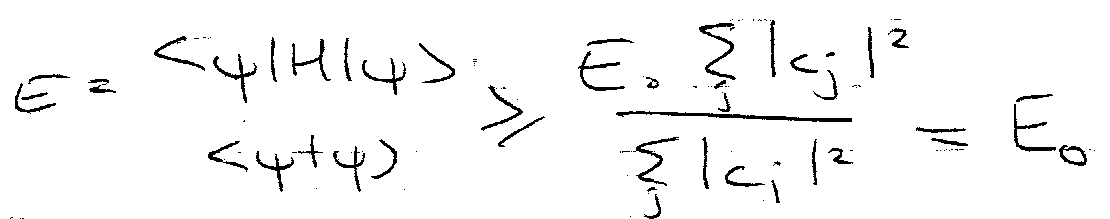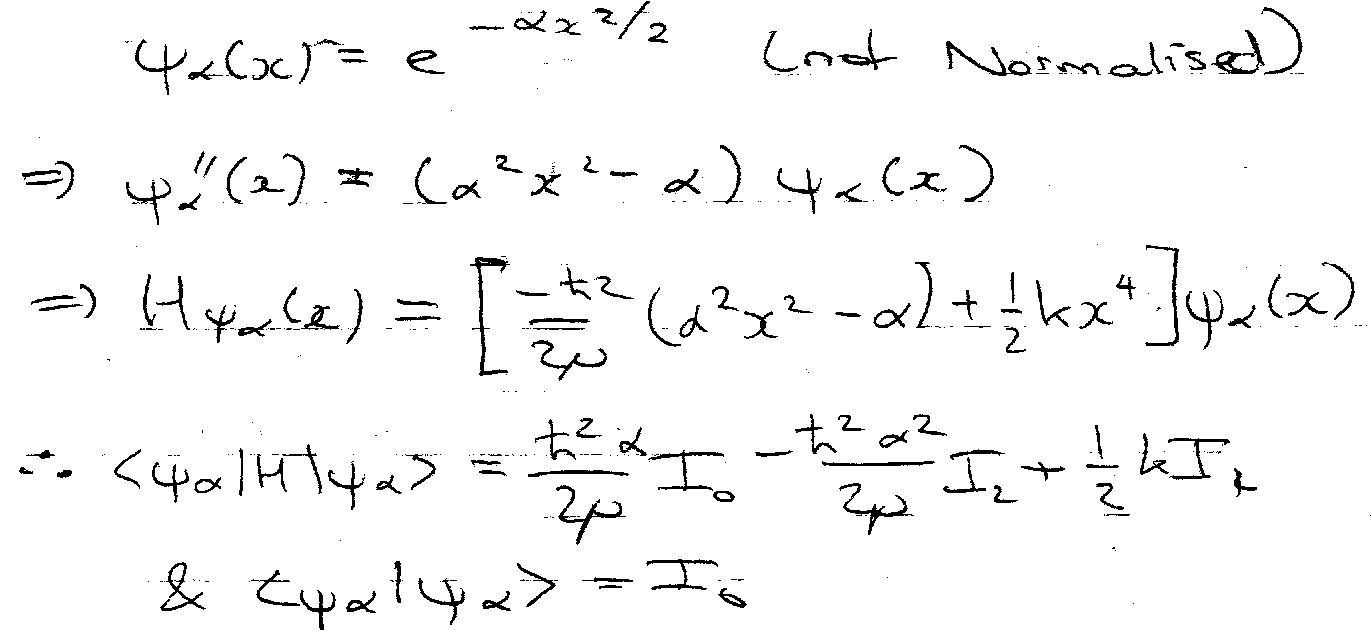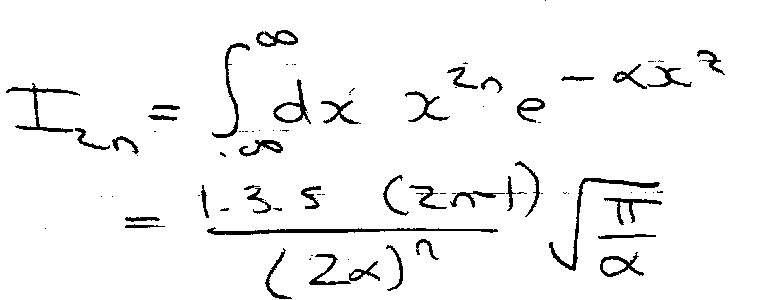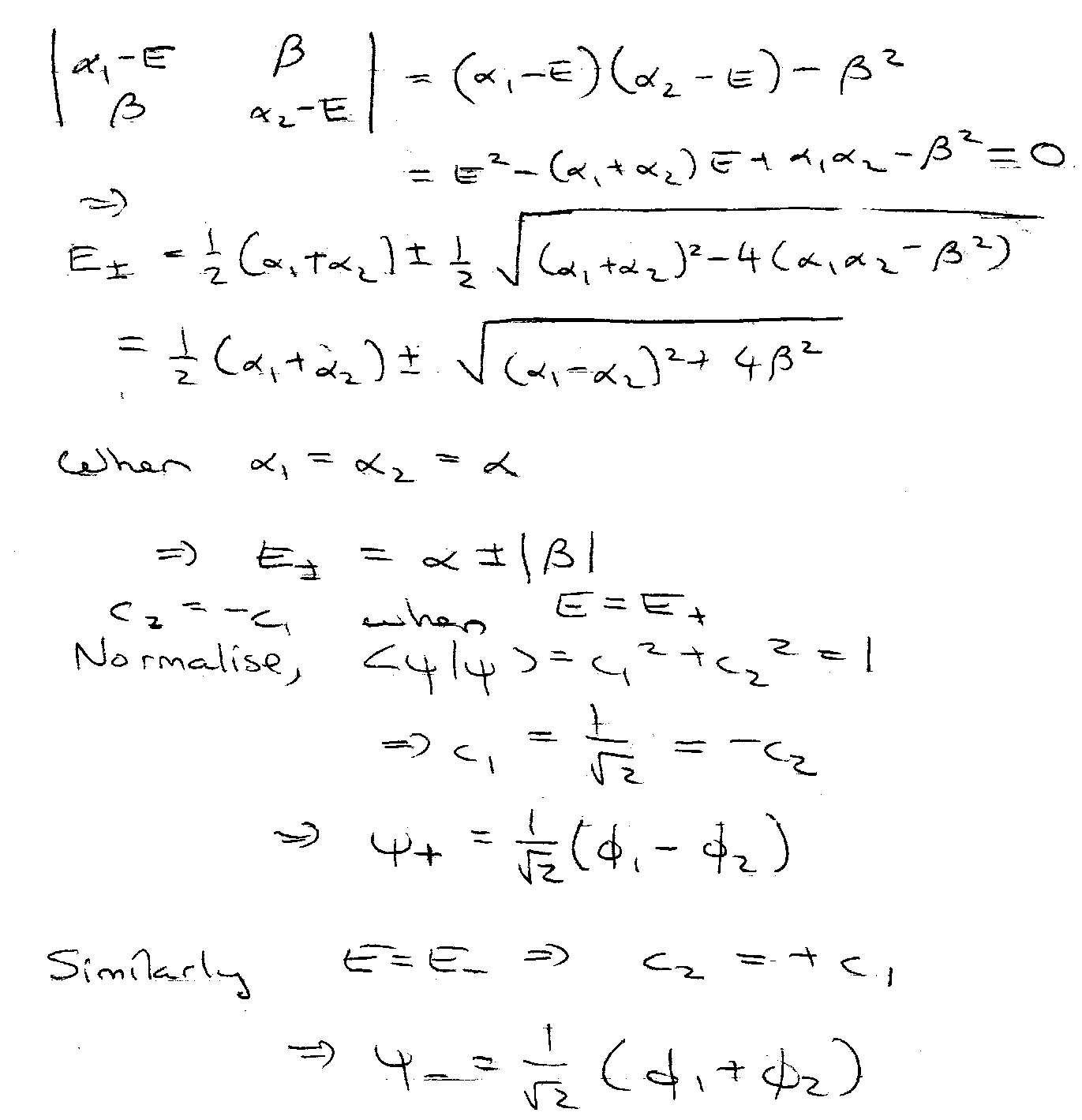Quantum Mechanics
Covers basics and postulates, before moving onto applications, e.g. particle in a box, on a ring, etc etc.
Quantum Mechanics Notes
The Basics of Quantum Mechanics
Schrödinger
The Born Interpretation
ψ at a point x, has a probability for a particle being between x and x+dx proportional to |ψ|2dx. Therefore,
Ωψ = ωψ
Ω = eigenvalue and ψ = eigenfunction.
When not an eigenfunction, must be a superposition or more than one wave function.
Particle in a Box
Tunnelling – possibility of finding particle outside box classically forbidden.
Vibrational Motion
Rotational Motion
Spectroscopy
Hydrogenics
Spin Orbit Coupling
Electron has spin angular momentum, and this generates a magnetic field.
Spin + orbit interactions – j = l ± ½
Multiplicity = 2S + 1
Postulates of Quantum Mechanics
1a: the state of a system of N particles is fully described by a function ψ(r1, r2, … rN; t) – the wavefunction.
NOTES:
Spin omitted (for now).
1b: Born’s Probabilistic Interpretation.
The probability that a system in a state ψ will be found in the volume element dτ = dr1, dr2 .. drN is ψ*(r1 … rN, t) ψ(r1 … rN, t) dτ.
NOTES:
Statistical, even for 1 particle.
P(x,t) = | ψ(x,t) | 2 is the probability density.
Can deduce from here:
Normalisation –
Conserves probability.
Physically acceptable ψ:
- Single valued.
- Continuous.
- Finite.
Dirac’s Bra-c-ket Notation –
2a: in quantum mechanics, observables are represented mathematically by operators: corresponding to every classical observable A there is a corresponding operator  which is linear and hermitian.
NOTES: e.g. x, px, E, etc.
3: Measurement. When a system is in a state described by ψ:
- single measurement of an observable A always yields a single result – an eigenvalue an of Â.
- Mean value of A equals the expectation value <Â>
Define Expectation Value:
[ if ψ is normalised ]
Bra-c-ket:
<Â> = <ψ|Â|ψ> if <ψ|ψ> = 1.
NOTES:
From (i) – Probability Distribution:
Common Sense, as mean A = sum over all n of Pnan.
From (ii) – must expand ψ in terms of eigenfunctions of Â.
i.e. Pn = | cn | 2
Probability Pn that particular value an is measures is |cn|2, where cn is the coefficient of eigenfunction fn of  (in the expansion).
Dispersion in distribution of measurements is characterised by:
Root mean square deviation (RMS) ΔA =
(ΔA)2 = < (Â - <Â>)2> = < (Â2 - 2Â <Â> + <Â>2 ) >
(ΔA)2 = <Â2> - <Â>2
Special Case –
ψ is an eigenfunction of Â, so Âψ = aψ. < Â > is:
< Â > =
Pn = 1 or 0 (n=ε or n≠ε), then dispersion free – single measure value, aε. < Â > = aε, < Â2 > = aε2, ΔA = 0.
Thus, if ψ is an eigenfunction of  then observable A will always yield the same result.
2b: Choice of Operators.
|
OBSERVABLE |
OPERATOR |
|
Position, x |
|
|
Linear momentum, px |
|
|
Total Energy, E |
|
All linear and hermitian.
Linearity –
Hermiticity –
 is Hermitian if: < m |  | n > = < n |  | m >*
EXAMPLES:
The eigenvalues of Hermitian Operators are real:
Orthonormality –
The eigenfunctions for different eigenvalues of Hermitian Operators are orthogonal, i.e. if  | fn > = an | fn > and  | fm > = am | fm >, where am ≠ an, then < fm | fn > = 0.
Time Dependence and Stationary States
Classical Observable → Quantum Operator.
e.g. Kinetic Energy,
T = ½ m (px2 + py2 + pz2) →
Potential Energy,
V = V(x,y,z), so
Total Energy = T + V, → H(x,px) [ Hamilton’s Function ]
Also note that the total energy operator from P2b is .
- Time Dependent Schrodinger Equation, TDSE.
H is independent of time (in conservative systems), so always separable solutions to the TDSE of the form:
A system in a state described by this is said to be in a stationary state.
Its energy is a precise quantity (P3) and no measurable property of the system changes with time, i.e. <A>(t) = <A>(0) where <A>(t) is the expectation value of operator A at time t.
Proof:
NOTES:
This resolves the “radiation paradox” of old Quantum Mechanics. Stationary State → still not solved the TDSE. Need φn from TISE.
A common relation that is useful is:
To use this, it is necessary to understand what a commutator is.
Commutators, Complementary Observables,
and the Heisenberg Uncertainty Principle
ABf ≠ BAf in general, where A and B are operators.
Define:
Commutator [A,B] of A & B:
[A,B] = AB – BA
Compared the effects on a ghost function, e.g.
[x, px] f = (xpx – pxx) f = -ih (x(d/dx) – (d/dx)x) f = -ih (x(df/dx) – (d/dx)(xf)) = ihf
Therefore [x,px] = ih.
If [A,B] = 0, they are said to commute. For example, [y,px] = 0 (independent x,y).
NOTES:
- [B,A] = - [A,B]
- [A,αB] = α[A,B]
- [A,B+C] = [A,B] + [A,C]
- [A,BC] = [A,B]C + B[A,C]
Uses:
From P3, Aψ = aψ and Bψ = bψ. This implies precise measurement of A and B, therefore [A,B] = 0 → precise value for each observable can be known simultaneously.
If [A,B] = 0, then there is an eigenfunction of A which is simultaneously an eigenfunction of B.
If [A,B] ≠ 0, it is NOT generally possible to measure the observables precisely and simultaneously.
These observables are said to be complementary or conjugate.
Heisenberg’s Uncertainty Principle:
ΔAΔB ≥ ½ | <[A,B] > |
Where,
(ΔA)2 = <Â2> - <Â>2
e.g. [x,px] = ih, → < [x,px] > = ih, so | < [x,px] > | = h
Hence, ΔxΔpx ≥ h/2
Applications of Quantum Mechanics
1 D free particle (no Potential)
Note that general solution to (1) is a linear combination:
General interpretation using P3 is that there is always dispersion-free energy for the above, but the relative probability of finding the particle moving in a given direction with momentum ±hk is |A’|2|B’|2
General Solution to the Schrödinger Equation from the above (2) & (3):
Compare to classical standing waves,
cos (2πx/λ) + sin (2πx/λ)
Therefore 2π/λ = p/h , → λ = h/p [ De Broglie Relation ]
Quantisation – Particle in a Box
Outside the Box: φ(x) = 0 – no particle here.
Inside the Box: V(x) = 0 so TISE same as free particle above. Solution as above is:
φ(x) = A cos (px/h) + B sin (px/h)
ψ(x) is continuous, therefore Boundary Condition: φ(0) = 0 = φ(L).
Hence,
φ(0) = 0 → A = 0.
φ(L) = 0 → B sin (pL/h) = 0.
Thus,
pL/h = nπ, where n = 1,2,3…
Also, p = √(2mE):
En = , n = 1,2,3…
Quantisation arose from the Boundary Condition. Quantum number n is established.
H φn(x) = En φn(x).
φn(x) = B sin (nπx/L) for 0 ≤ x ≤ L
φn(x) = 0 for x ≤ L, or x ≥ L
B, such that φn(x) is normalised, is found by:
Pictorially,
Probability Density | φn(x) | 2 → tends to classical limit (Correspondence Principle)
Energy Level Separation:
ΔE = En+1 – En =
So, ΔE → 0 as L → ∞
Extra Dimensions:
d = 2, En =
Lx = Ly → square box. n1 = n2 is single degenerate, while n1 ≠ n2 is doubly degenerate.
Degeneracy is a consequence of symmetry.
Harmonic Oscillator
Classically, E = T + V = px2/2μ + ½ kx2
Quantum Mechanics:
E =
Satisfied for all x if both:
Normalise,
For remaining solutions try:
Pn(x) = polynomial in x.
Gives second order differential equations for Pn(x) when subbed into the Schrodinger Equation.
The solutions are called Hermite Polynomials.
Pn(x) = Hn (α x)
Eigenvalues En = (n+½)hω , n = 0,1,2…
Ho (α x) = 1 [ even in x ]
H1 (α x) = 2α x [ odd in x ]
H2 (α x) = 4αx2 – 2 [ even in x ]
… etc.
Hence,
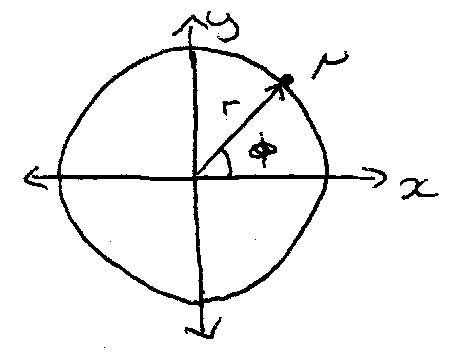
Particle on a Ring
To get H:
Transform to polar cords.
Fix r, look at angular component of H.
Transforming to polar coordinates:
Fixing r means that & can be dropped. ψ = ψ(φ): dψ/dr = 0 = d2ψ/dr2.
Thus,
Therefore eigenfunctions of H can be chosen to be eigenfunctions of Lz.
Then,
Therefore normalised eigenfunctions of H = which satisfy the boundary condition are:
NB: Em = is doubly degenerate for | m | > 0.
Particle on a Sphere
Transform to polar cords.
Consider angular parts.
Separate θ/φ dependence.
Separating variables,
Ordinary Differential Equation for (H)(θ), the Legendre Equation.
Solutions are associated Legendre functions,
i.e. m = -l, -(l-1), … 0, … (l-1), l.
This gives the spherical harmonics:
Which satisfy:
Molecular Rotation
Equivalent to free motion of particle with reduced mass on surface of a sphere (radius re).
Therefore H = [ J not L – convention for molecular systems ]
i.e. EJ = Bhc J(J+1) [ rotational energy levels ]
B = h/(8π2Ic) [ units m-1 or cm-1 ]
J = 0,1,2 …
M = -J, -(J-1) … J. [ projection of momentum along z ]
This is (2J+1) degenerate.
Atomic Orbitals
First, it is useful to refine our units onto the atomic scale:
In atomic units,
From (2) and (3), any atomic orbital can be written as separable:
This is the “associated Laguerre Equation”. En = -1/(2n2) Hartree, n = 1,2,3 …
Electron Spin
Stern & Gerlach (1922) passed beam of Ag atoms through an inhomogeneous magnetic field (i.e. a field gradient was present → force). Beam split in two.
Ag (5s1). If S = ½, then ms = ± ½ … 2 components with different energies in a magnetic field.
1925 – splittings in atomic spectra. e- had intrinsic angular momentum of ½ h.
1930 – Dirac. Obtained wave equation for e- by combining Quantum Mechanics and Special Relativity. Equation predicted s = ½, confirming the above.
E = - μ . B
If B is in the z-direction, s.B = szBz, →
Spin Wavefunctions
Single electron:
Satisfy usual angular momentum eigenvalue equations:
Also,
Two electrons:
Only linear combinations of following possibilities –
Possible values of Ms = m1 + m2 are:
Ms = 1,0,-1 (S=1) and Ms = 0 (S=0).
MS = S, S-1, .. –S → (2S+1) = 3 (triplet).
MS = S, S-1, .. –S → (2S+1) = 1 (singlet).
But what are corresponding 2 electron spin wavefunctions, | s, ms > ?
| S = 1, Ms = +1 > = | α1 α2 >
(as this is the only way to get Ms = +1).
Similarly,
| S = 1, Ms = -1 > = | β1 β2 >
(Ms = -1).
Both of these S=1 wavefunctions are symmetric wrt interchange of the 2 electrons (e1↔e2).
Hence, the remaining S=1, Ms=0 component of the triplet states must be symmetric too [ Ms quantum number depends on where we choose to put the z-axis, which clearly cannot affect the exchange symmetry of a triplet state ].
Hence, only possibility for:
| S = 1, Ms = 0 > = symmetric combination of , where the term outside the brackets is a Normalisation Constant.
Similarly,
| S = 0, Ms = 0 > = , which has antisymmetric exchange symmetry.
Pauli Exclusion Principle
Suppose quantum system contains two indistinguishable particles 1 and 2 such that:
ψ = ψ(1,2)
i.e. ψ is a function of all space and spin coordinates.
Repeat operation:
Corollary 1: Exclusion Principle in Orbital Space.
No 2 electrons can have the same set of 4 quantum number (n,l,ml,ms) within the orbital approximation.
Combination with the Aufbau Principle gives the Periodic Table.
Reason:
ψ(2,1) = ψn,l, ml, ms(2) ψn,l,ml,ms(1) = ψn,l, ml, ms(1) ψn,l,ml,ms(2) = +ψ(1,2)
But electrons are fermions so ψ(1,2) = -ψ(2,1).
Corollary 2: Exclusion Principle in Real Space.
2 electrons in a triplet state (S=1) cannot be at the same point in space.
ψ(1,2) = ψspace x ψspin
ψ(1,2) = ψ(r1,r2) x
Thus, ψspace must be antisymmetric wrt e1↔e2.
ψ(r2,r1) = - ψ(r1,r2)
Setting r2 = r1 = r:
ψ(r,r) = - ψ(r,r)
Therefore ψ(r,r) = 0 and | ψ(r,r) |2 = 0, so get a “Fermi Hole”.
Basis of Hund’s 1st Rule: triplet states are lower in energy than singlet states, all other things – including the electron configurations – being equal.
The Variational Method
How to find good approximate solutions to problems that can’t be solved exactly.
Where ψ = trial wavefunction and Eo = exact ground state energy.
So,
Suppose we have a trial wavefunction ψα that depends on a parameter α. Find the best α (most accurate wavefunction) by minimising:
Example – Quartic Oscillator
Similar to Harmonic Oscillator, so try that wavefunction as trial:
Where,
Hence,
Example 2 – Secular Equations
I am happy for them to be reproduced, but please include credit to this website if you're putting them somewhere public please!