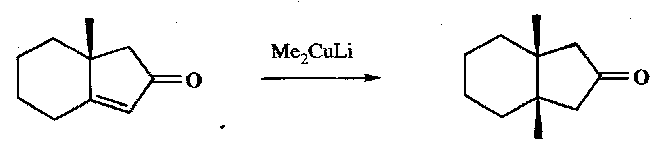Organometallics
Divided into saturated, unsaturated and aromatic metallated compounds. Fairly simple, but useful for going over pre-Finals.
Organometallics Notes
Metallated Saturated Hydrocarbons
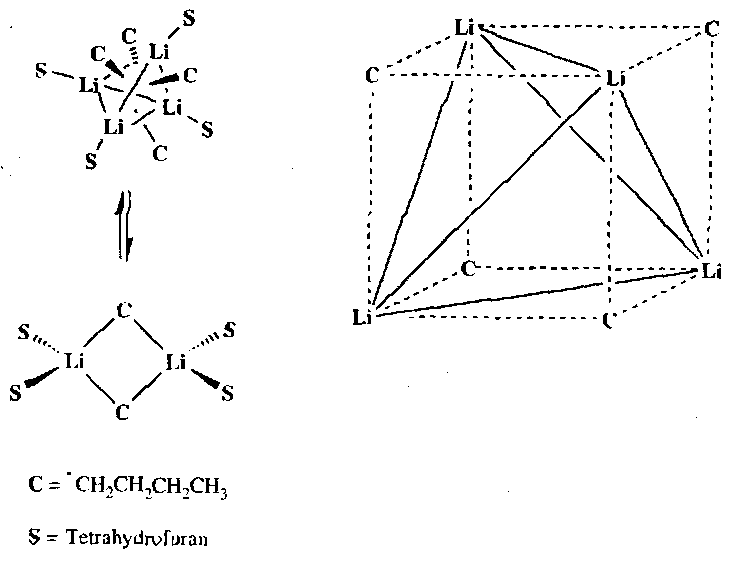
Lithium Reagents
BuBr + 2Li → BuLi + LiBr, in Et2O @ -20oC.
Framework Structure –
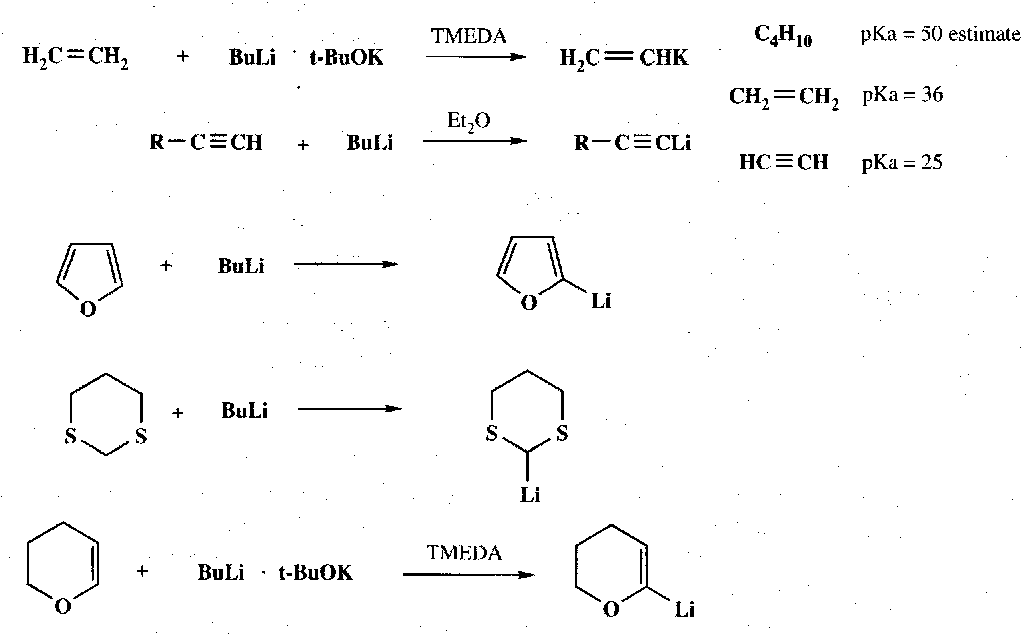
Main use is as a strong base. This is enhanced by co-solvents such as HMPA, which coordinates the Li leaving monomeric Bu-.
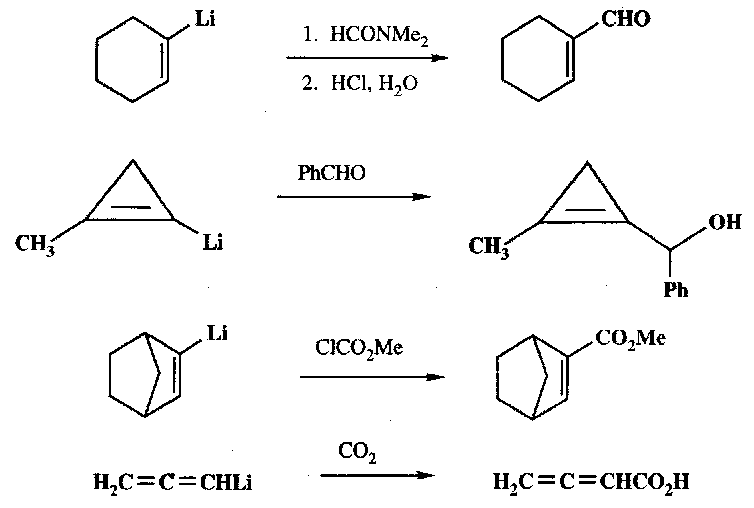
Examples –
Grignard Reagents
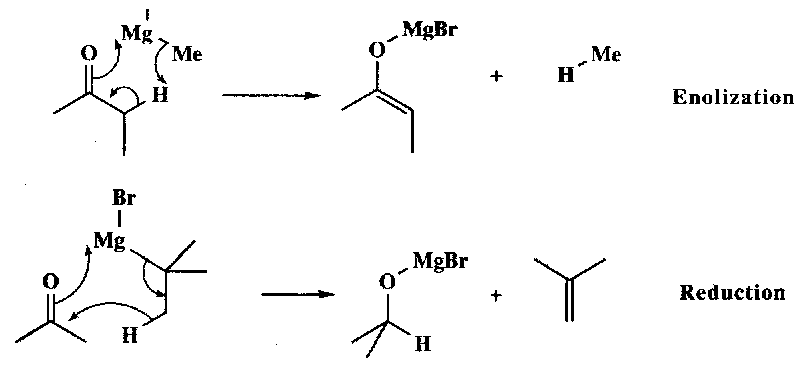
These are both basic and nucleophilic. Reactivity depends on the Schlenk Equilibrium:
2MeMgBr ⇌ Me2Mg + MgBr2
MgBr2 coordinates to E+, making it more susceptible to attack by nucleophilic Me-.
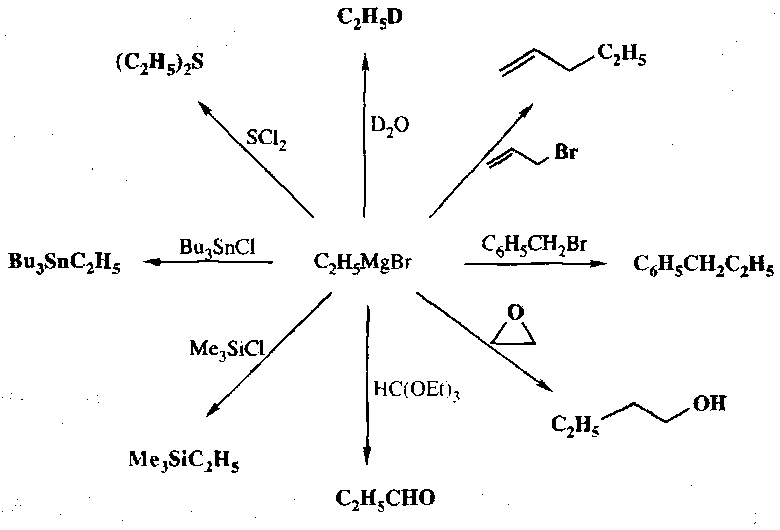
Metallated alkanes attack carbonyls readily, forming the alcohol after workup. Side reactions do tend to occur, for example deprotonation α to C=O, or β-H transfer (if the organometallic reagent has one).
Stereochemistry
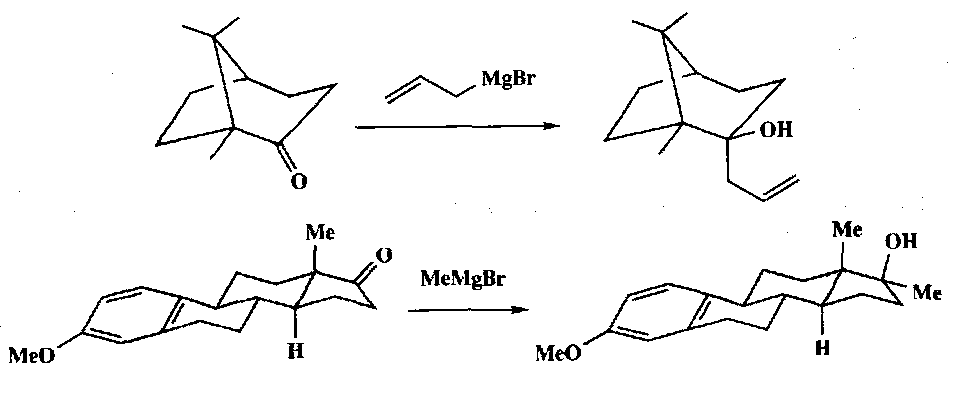
In conformationally locked ketones the less hindered face of the carbonyl group is attacked:
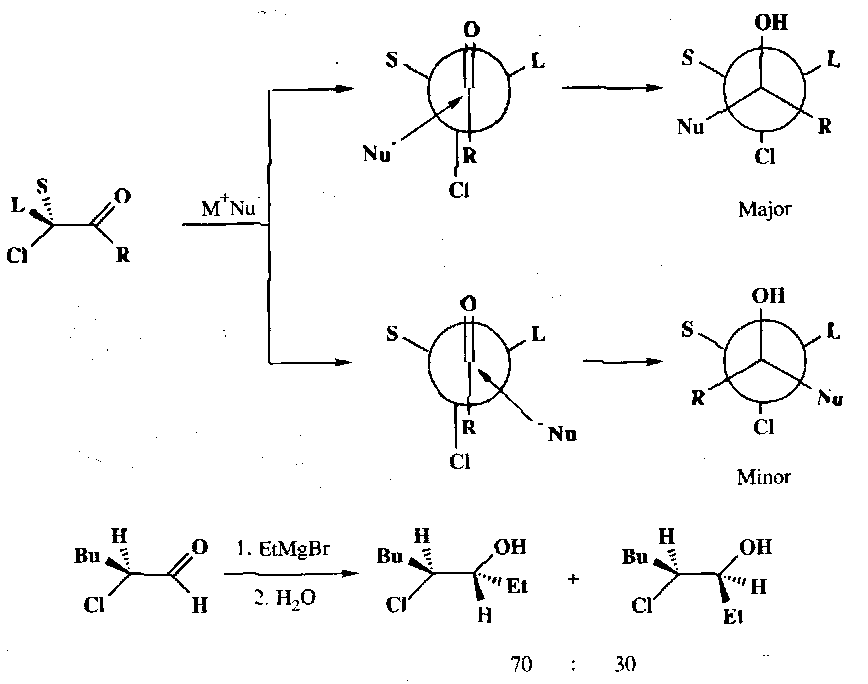
The two faces of a carbonyl compound with an α-chiral centre are diastereotopic and hence nucleophilic addition to such compounds can lead to two diastereomeric products. Cram’s Rule is a way of predicting which of the two faces is preferentially attacked.
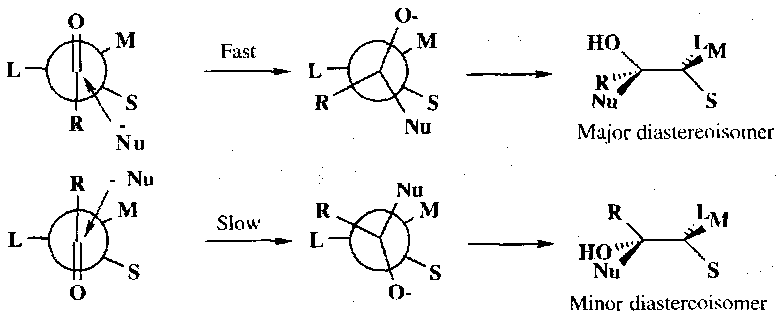
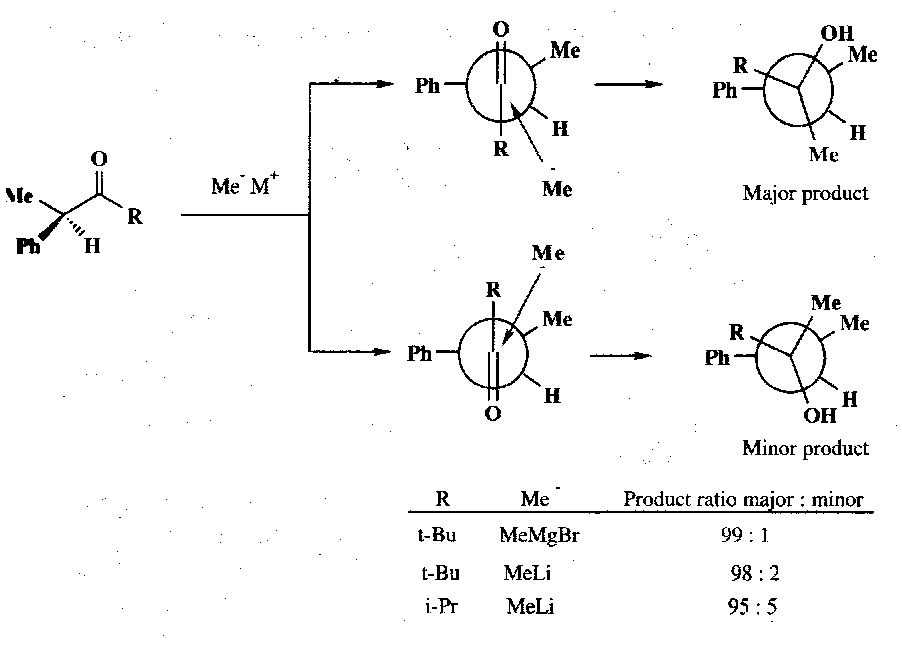
Although there is free rotation about the single bond between the carbonyl and the α-carbon, when applying Cram’s Rule we say that certain conformations will be more reactive and these will be used to predict the major product. When the α-carbon has three groups which may be classified as small (S), medium (M) and large (L), then the most reactive conformation is where the groups are staggered such that the carbonyl oxygen is between the large and medium groups (Felkin-Anh). Nucleophilic attack occurs anti to the large group to give the major product diastereoisomer. The other face of the carbonyl is attacked in a more hindered, staggered conformation in which the carbonyl oxygen lies between a large and a small group, leading to the minor isomer:
In these two conformations the most important steric interactions are determined by the R group which prefers to lie between the large and small groups rather than between the large and medium groups:
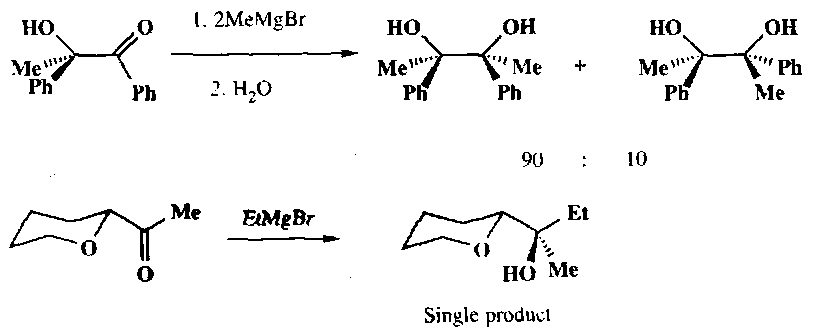
Attack on a different conformation may be preferred when one of the substituents on the α-carbon has a lone pair, which may coordinate to the organometallic reagent. Here the carbonyl group and the coordinating substituent are held syn-periplanar by a 5-membered chelate ring. Attack by the nucleophilic then occur preferentially from the less hindered side:
This can give rise to particularly stereoselective reactions:
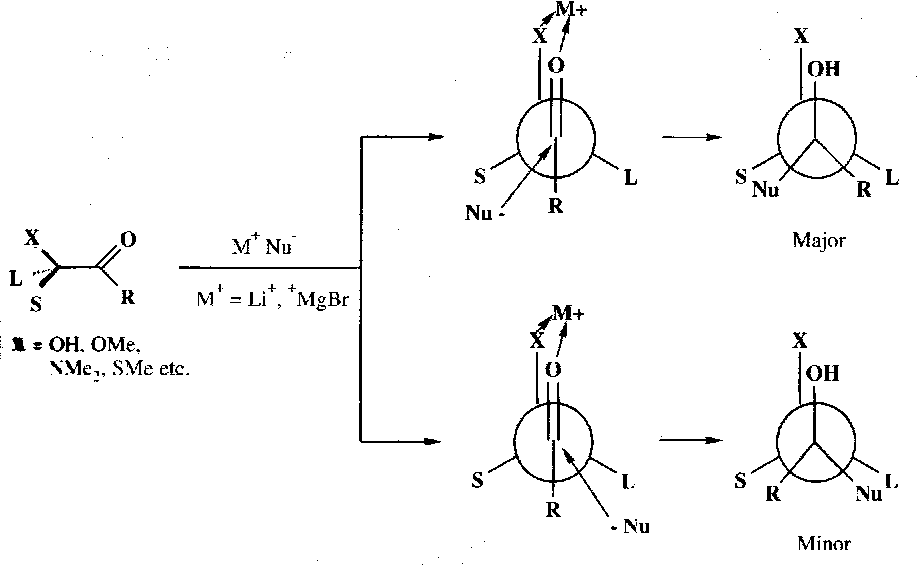
Preferentially nucleophilic attack on a third type of preferred conformation is encountered when the α-carbon has an electronegative substituent (e.g. halide). The most stable conformation places the dipoles of the carbon-oxygen and carbon-halide bonds antiperiplanar to one another and attack occurs on the less hindered side of the carbonyl group:
Other Reactions
They also attack esters and amides. One equivalent gives the ketone, but more allows further reaction to give the alcohol.
They will also attack CO2 (carboxylating the alkane), and similar attack CS2 and carbonates. Ketenes and isocyanates are also readily attacked, forming the ketone and secondary amide respectively.
Alkylation of metallated alkanes is complicated by metal exchange (especially for alkyl halides) and particularly subsequent elimination after attack.
Organocuprates
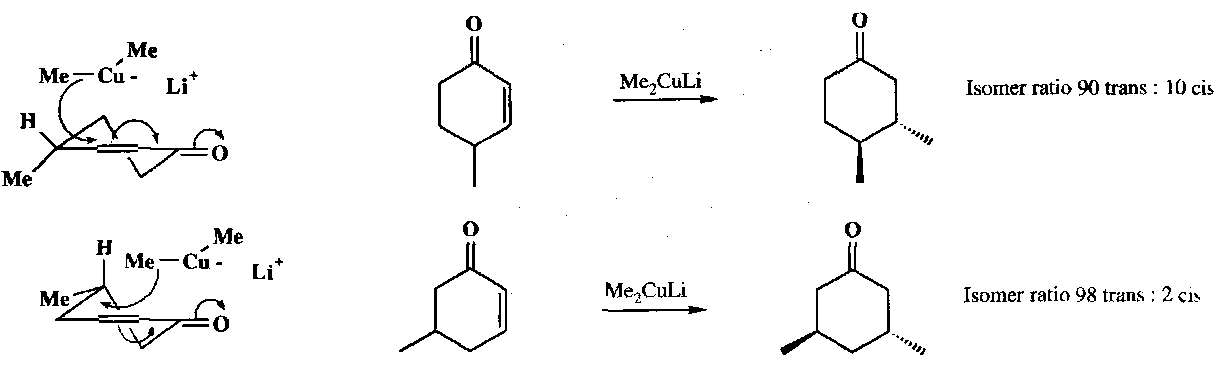
Also attack α,β-unsaturated ketones to give the enolate (i.e. 1,4 attack) – very useful.
Stereochemistry –
Also,
Cis product formed, as the cis-fused 5,6 ring is much more stable.
Organozinc & Organocadmium
These are both less reactive. They will not attack esters, and barely react with ketones (Zn slowly reacts with the latter, while Cd does not).
Metallated Alkenes & Alkynes
For Li, these are made by transmetallation, addition of BuLi (or similar), or the Shapiro (and similar) reactions. Grignards can be made by the same method as for alkanes. They both give rise to vinyl carbanion-like reagents.
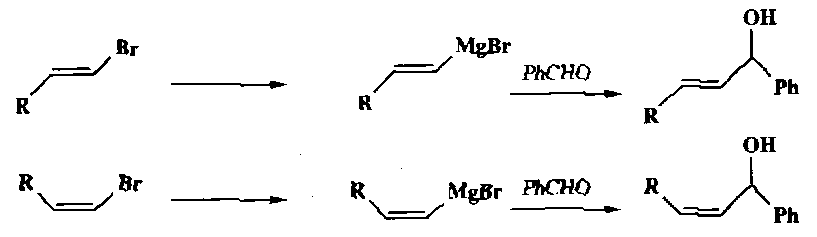
They are less basic than the alkanes, so are primarily nucleophiles. This is useful synthetically:
Note the retention of stereochemistry:
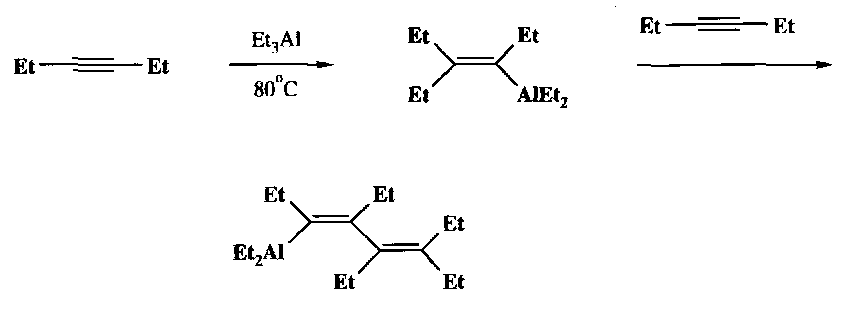
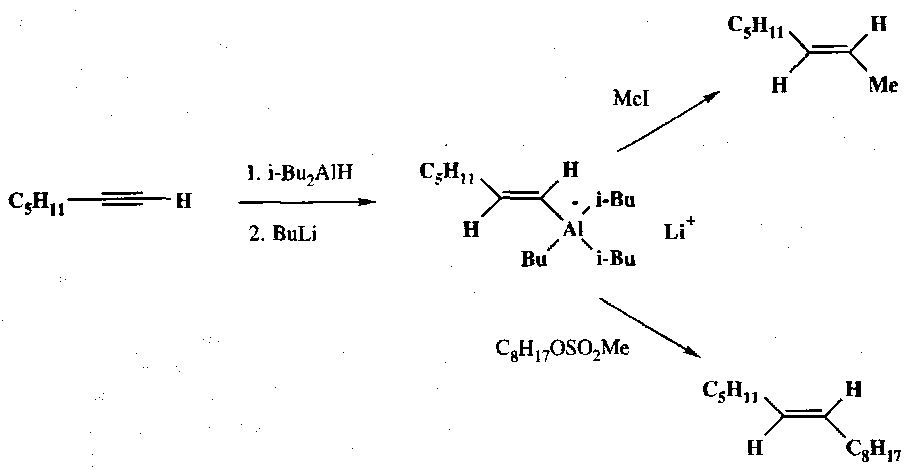
Vinylaluminium reagents are also widely used. They undergo stereoselective cis-addition of trialkylalanes with acetylene derivatives:
These then react in various ways, such as stereoselective protonation:
Similarly with halogens, to give vinyl halides.
They also undergo nucleophilic substitution:
Also, nucleophilic addition to carbonyls just like other organometallics (C=O more electrophilic than the halogens/halides above).
Metallated Alkynes are similarly (in fact more so) nucleophilic, and are useful for introducing a C≡C group into a molecule.
Metallated Aromatics & Heterocycles
Benzene can be metallated thermodynamically, but the reaction is usually very slow. Hence, these are most efficiently prepared from phenyl halides, in analogous manners to other organometallics.
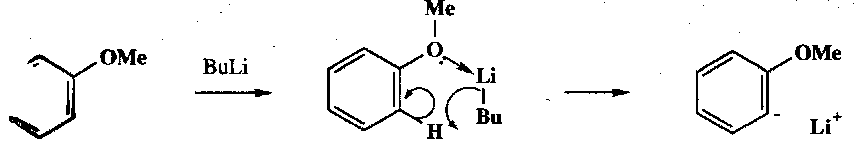
A particularly useful synthetic reaction is ortho-lithiation:
Electron withdrawing Y groups or those with lone pairs control the ortho stereochemistry by coordinating Li. It also helps if the Y group is stable wrt nucleophilic attack (e.g. sulphonamides).
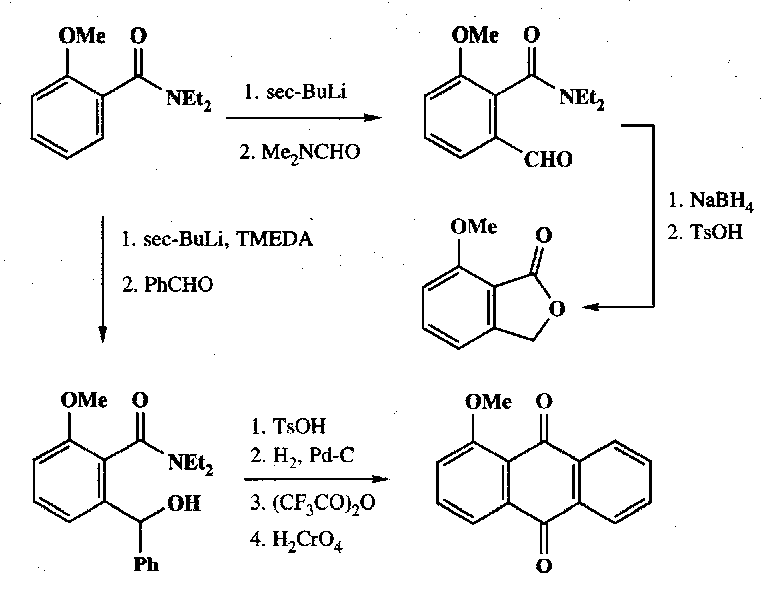
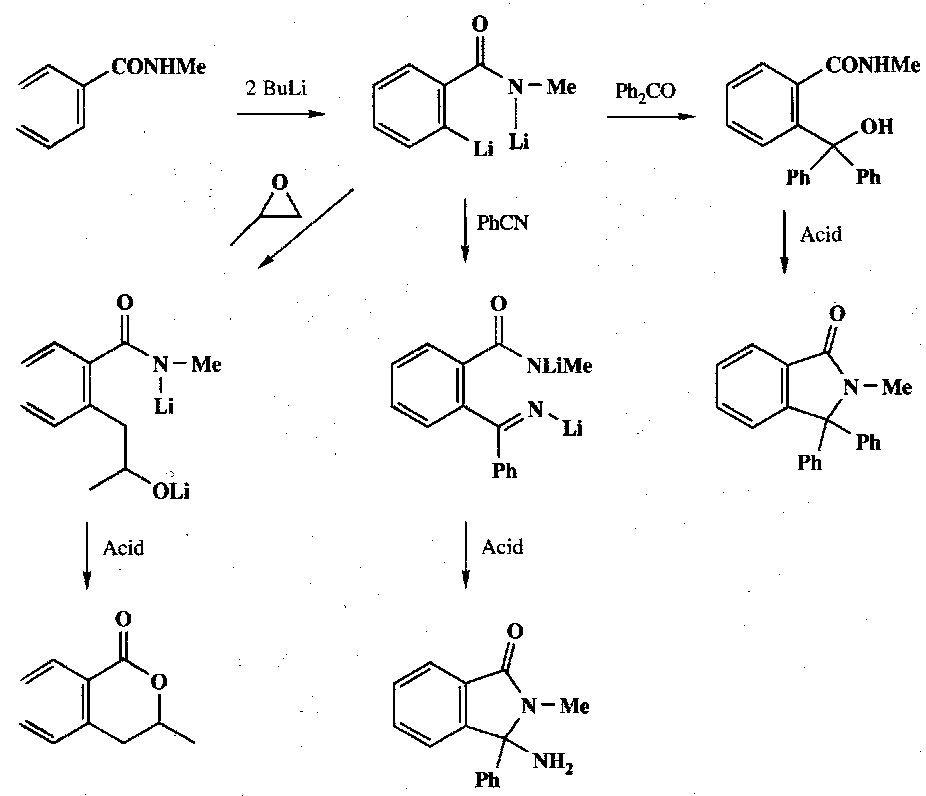
Examples –
Also, benzyne generation:
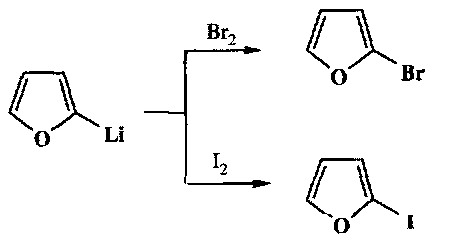
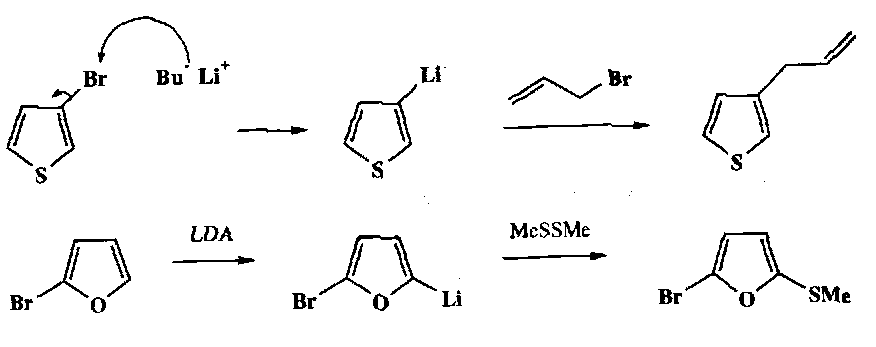
Heterocycles such as furans, pyrroles and thiophens can be directly metallated with the need for a halide substituent. This provides an easy way of attaching side chains to the 2-position (next to the heteroatom). Indoles etc. behave in a similar way (note that the for unsubstituted heterocycles, whatever the type, the most acidic H is always the position that is metallated).
If Li at the 3-position is needed, then a 3-substituted halide heterocycle is required, e.g.
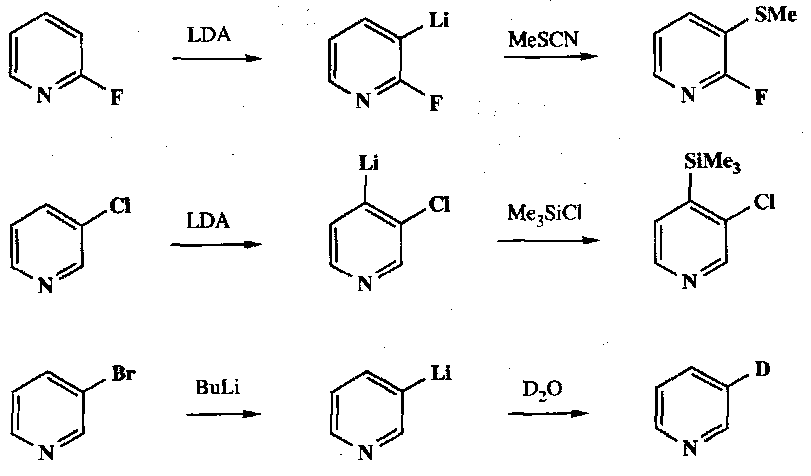
Note that Pyridine can be unpredictable in its metallation:
I am happy for them to be reproduced, but please include credit to this website if you're putting them somewhere public please!