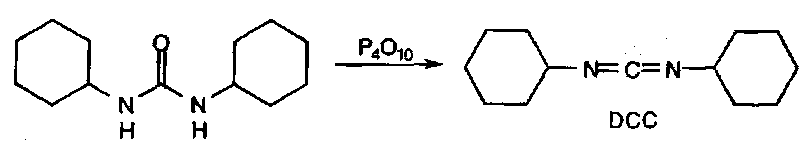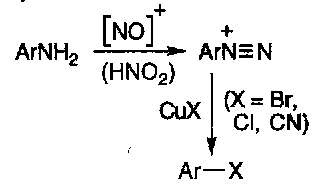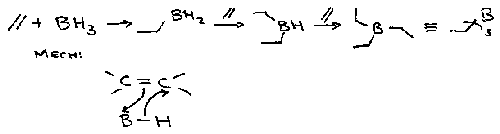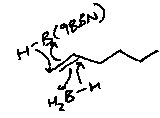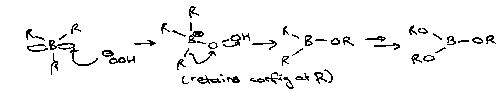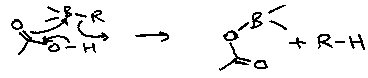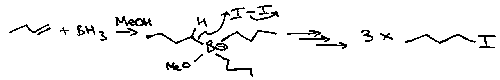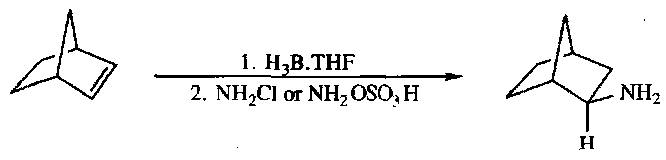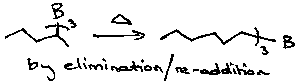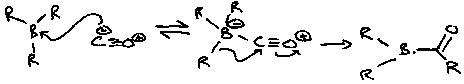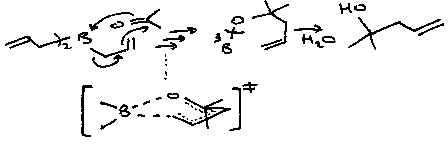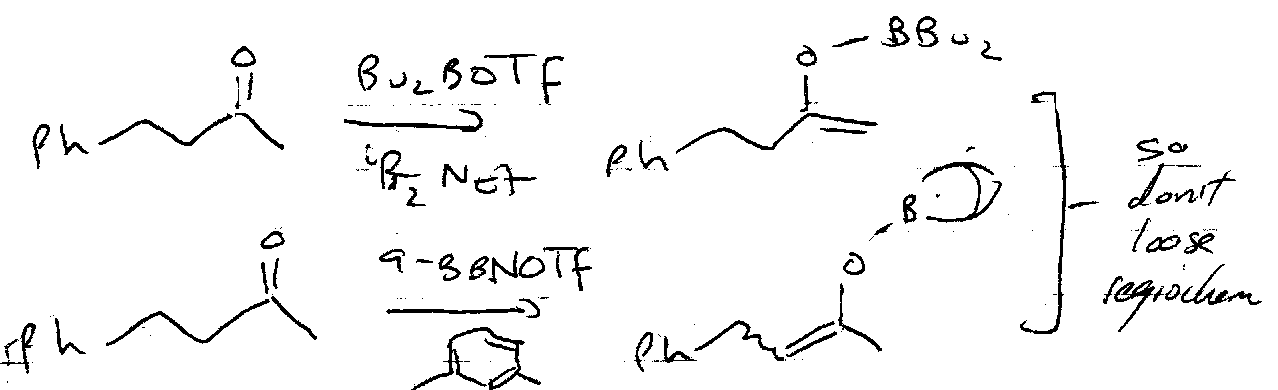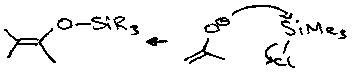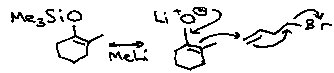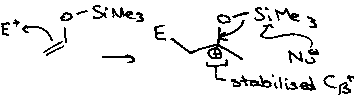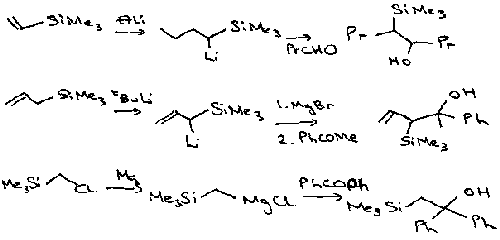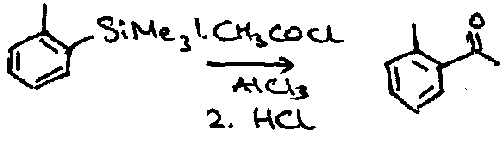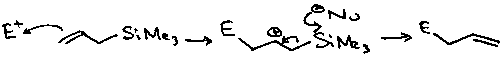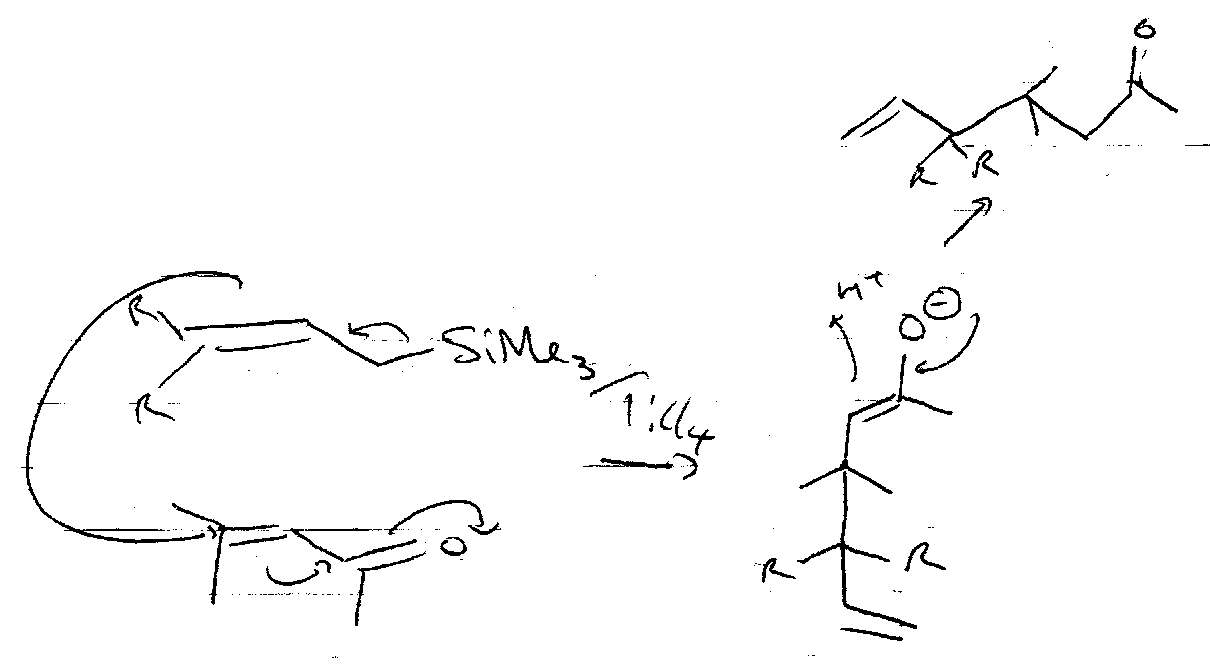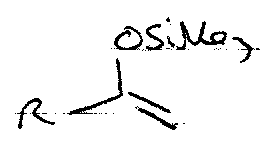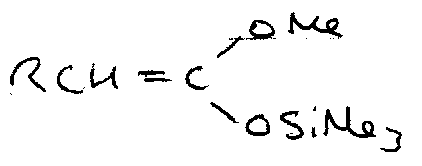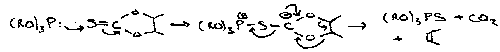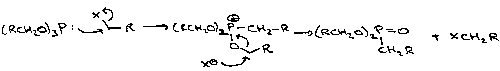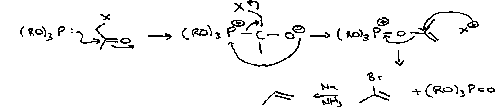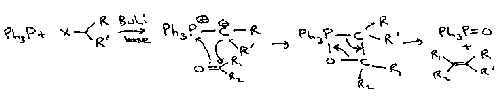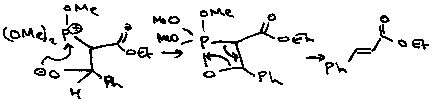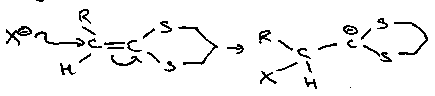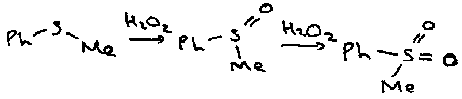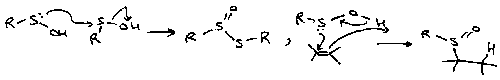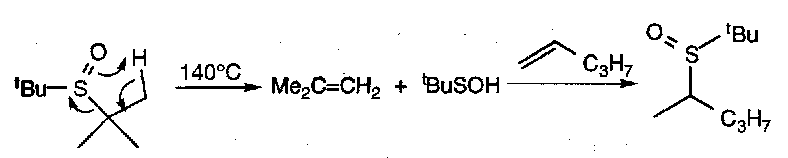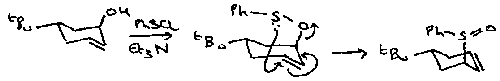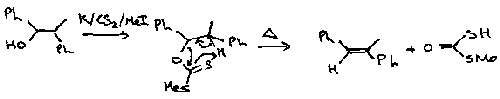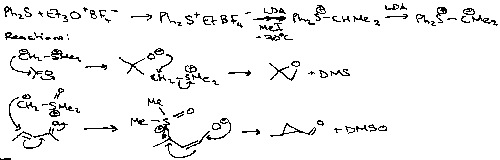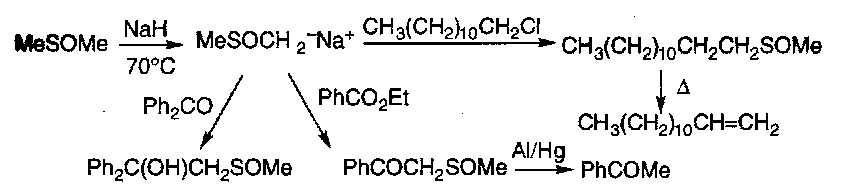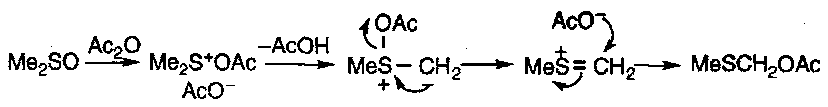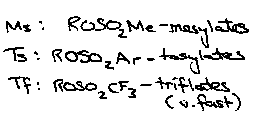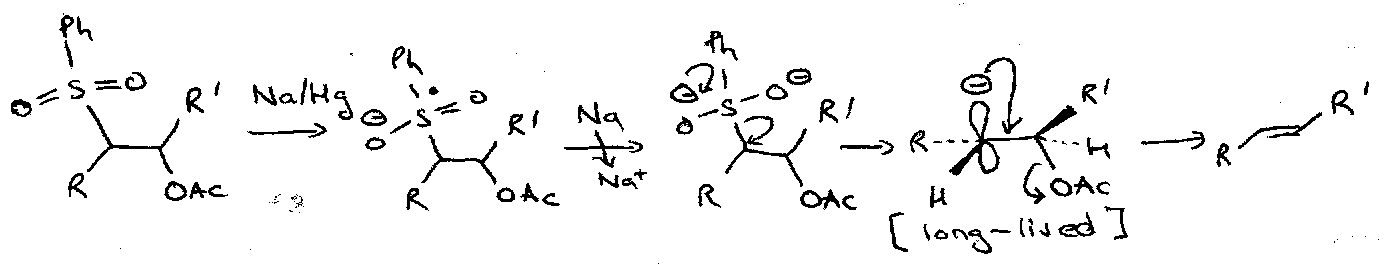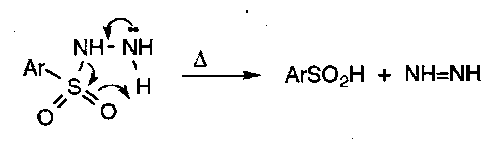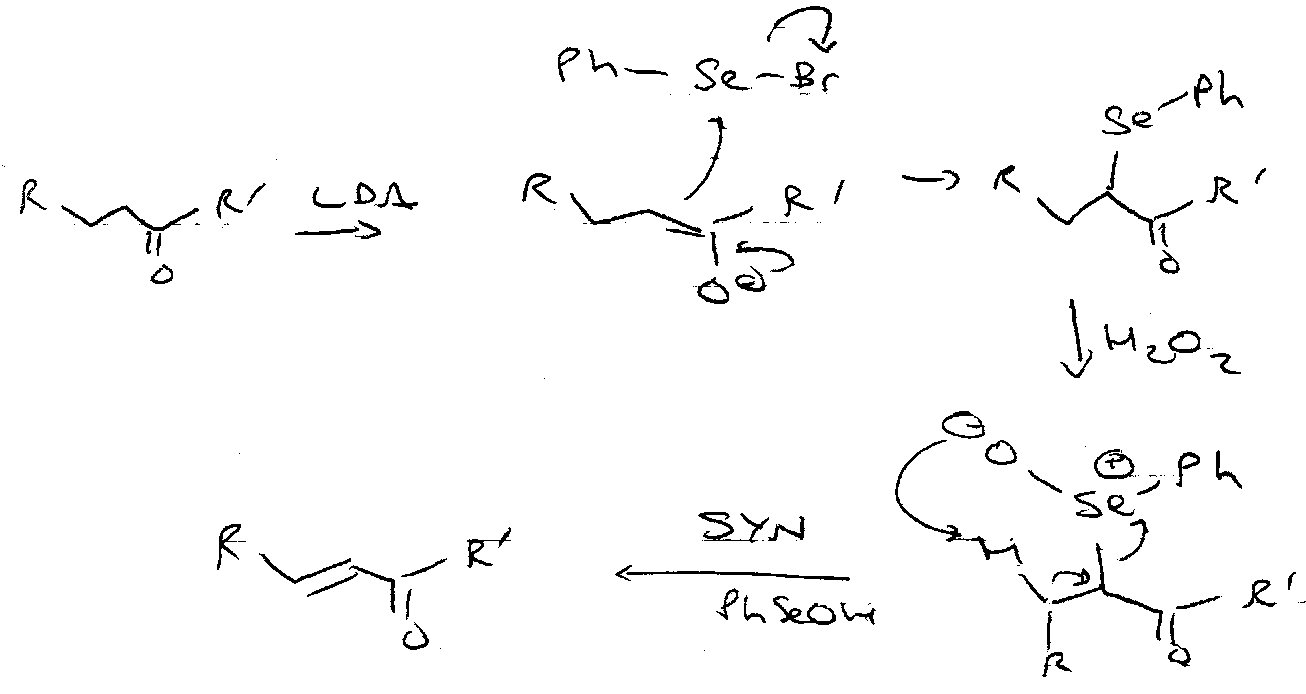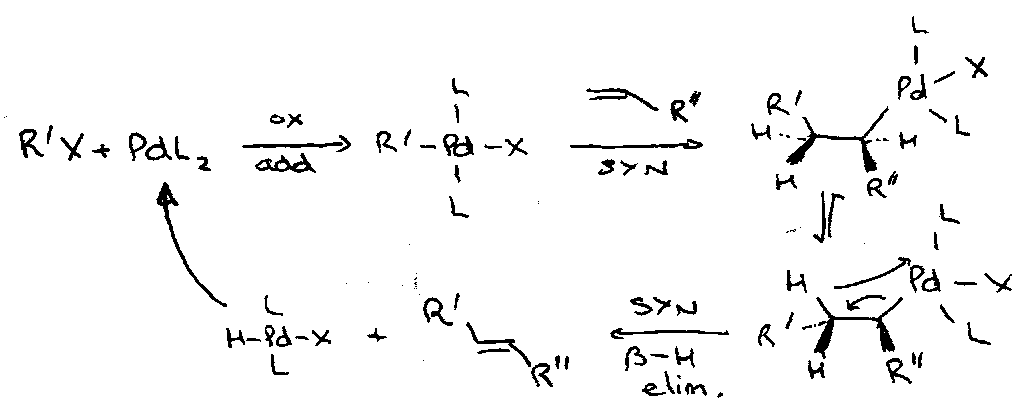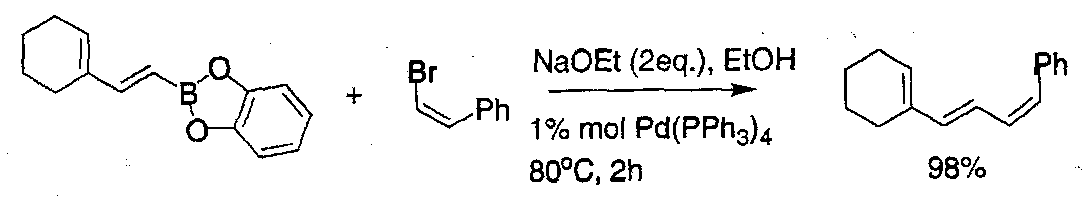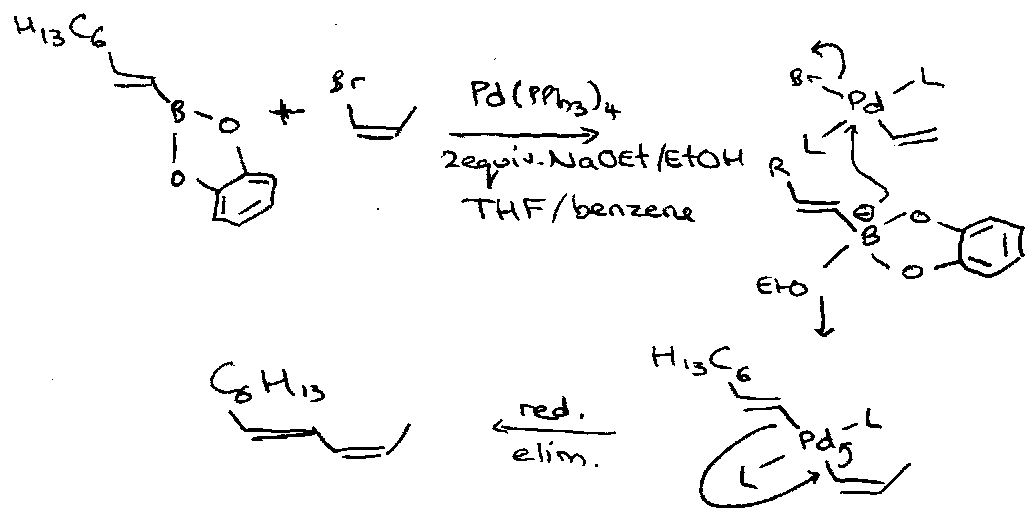Organoelements
Covers Organonitrogen, Organoboron, organosilicon, organophosphorus and organosulphur reactions and reagents. Mostly from the primers (where appropriate) but added to from core texts too.
OrganoElements Notes
NITROGEN
C-N σ-bonds are almost as strong as a typical C-C bond, so they are generally hard to break (usually need to be charged). They are, however, slightly polarised due to the electronegativity difference.
Much of the organic chemistry is driven by the lone pair on N. It also allows N to form tetravalent (positively charged species), which are common. Hence it is basic.
Amines
pKa of ammonia is 9.26. Substituting alkyl groups for H’s increases the pKa (i.e. are more basic). Electron-donating groups stabilise the positive charge, reducing Ka and therefore raising the pKa.
Obviously, they are also nucleophiles. Amines react with alkyl halides via an SN2 mechanism. We expect to see multiple nucleophilic substitutions as the amine gets more nucleophilic (alkyls e-donate to the N) such that we end up with tertiary amines or ammonium salts. Generally, secondary amines are the best nucleophiles (electrostatics) followed by primary (kinetics), with tertiary amines the worst nucleophiles (sterically hindered).
Amines also react with carbonyls, forming imines and enamines, and with nitrous acid (HNO2) – see later.
Synthesis of Amines
As has been mentioned earlier, forming amines by nucleophilic substitution with alkyl halides etc. is unlikely to be useful, as further substitutions are likely to occur. Most methods now use “monophilic” ammonia-based compounds.
Some examples:
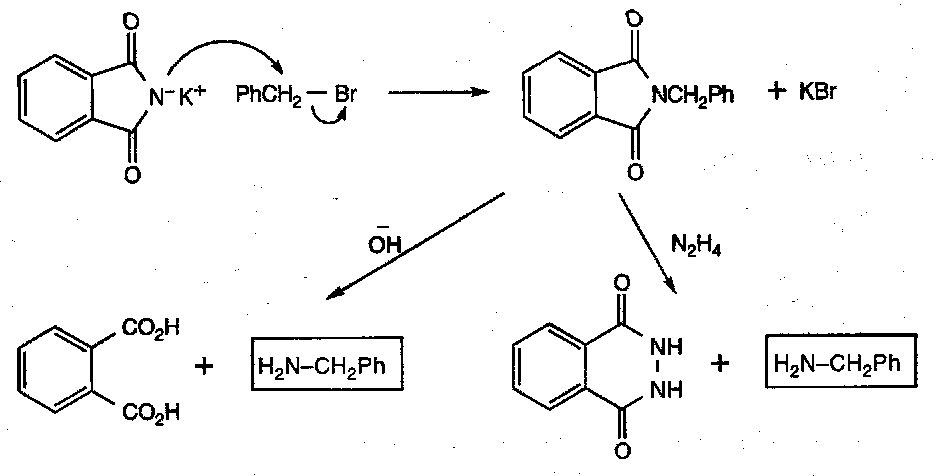
The Gabriel Method
Note that a modified version of this with malonic esters allows the synthesis of simple amino acids.
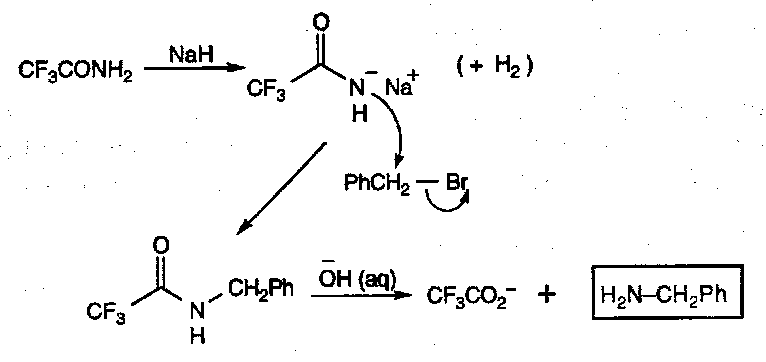
CF3CONH2/NaH, then OH-(aq)
Same type of reaction as the Gabriel method, but milder conditions.
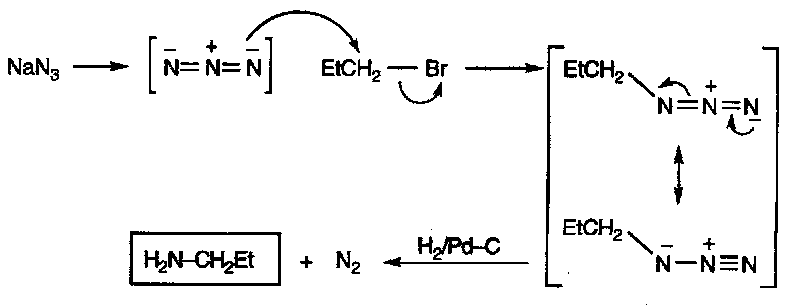
NaN3 then H2/Pd-C
This gives good yields. Azide is a good nucleophile that will only react once, and it is then reduced.
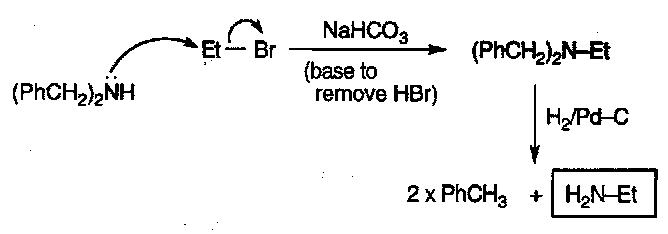
(PhCH2)2NH then H2/Pd-C
Based on the removal of the benzyl group by hydrogenation.
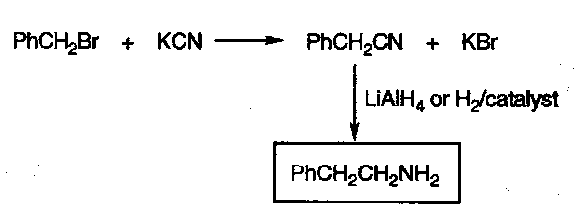
KCN, then reduction
Another nucleophile that reacts only once. The product gains an extra carbon atom, as the C is the nucleophilic part of the cyanide.
Amines via Reduction Reactions
|
Reagent |
LiAlH4 |
NaBH4 |
H2/catalyst |
|
Imines |
✓ |
✓ |
✓ |
|
Amides |
✓ |
✗ |
✗ |
|
Nitro Groups |
✓ |
✗ |
✓ |
|
Nitriles |
✓ |
✗ |
✓ |
Imines
R1R2C=N-R3
- Susceptible to nucleophilic attack on carbon.
- Easily protonated to produce iminium ions.
- Prepared by reacting an amine with an aldehyde or ketone (requires water removal to prevent equilibrium lying on amine + carbonyl side).
Reactions of Imines
It is always worth bearing in mind that imines and iminium ions are very easily hydrolysed, which can lead to useful workup, or require specific conditions to avoid it.
Imines are usually attacked by carbon nucleophiles, such as enolates and Grignards. For most nucleophiles, the imine is not trapped in situ. However, in the Mannich-type Reactions and also the Strecker Reaction it is.
Strecker Reaction:
Synthesis of amino acids.
Mannich Reactions:
Can trap iminium ions intermolecularly or intramolecularly.
e.g.
These reactions are successful because of the unhindered nature of the iminium ion, and also that it holds a full positive charge and is thus very reactive.
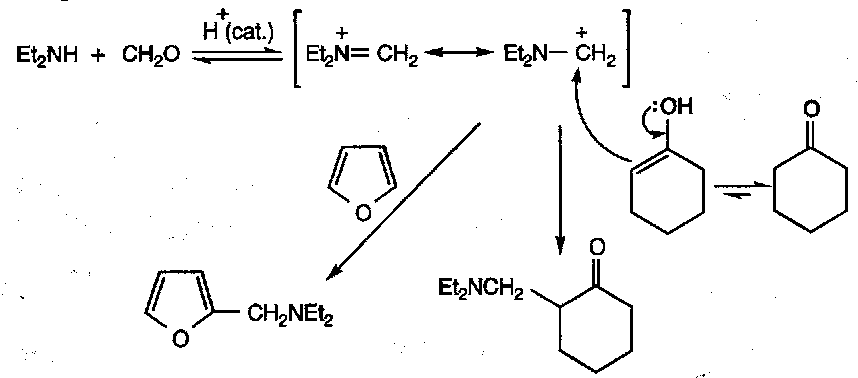
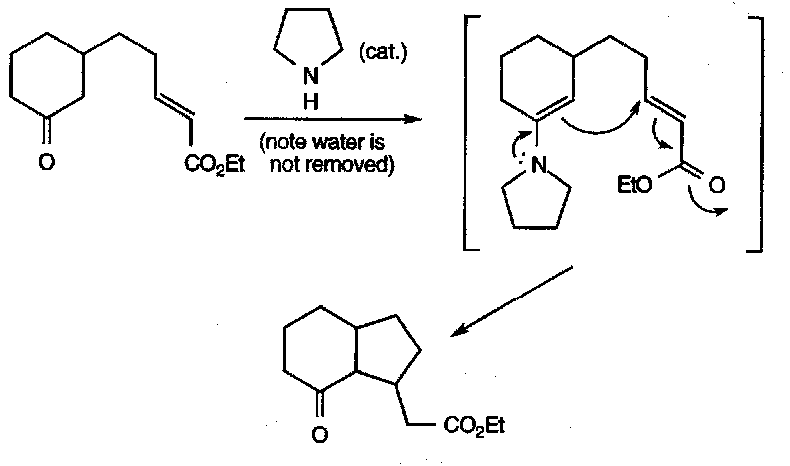
Intramolecular versions happen very readily, and often lead to cyclisations, e.g.
This reaction is common in biosynthesis of many alkaloids.
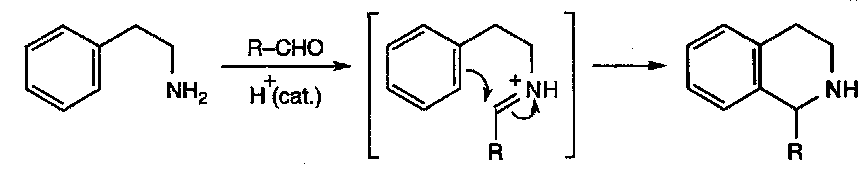
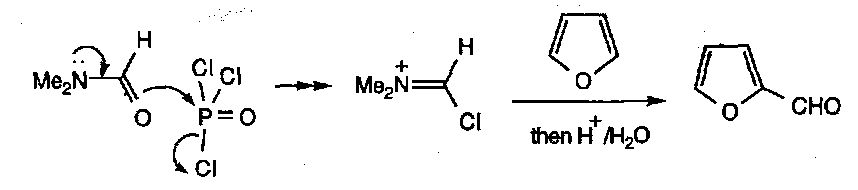
Vilsmeyer Reaction:
Extension of the Mannich Reaction:
Good for forming aldehydes.
Reduction of Imines
This is a simple process that yields amines. Typical reducing agents are catalytic hydrogenation or NaBH4.
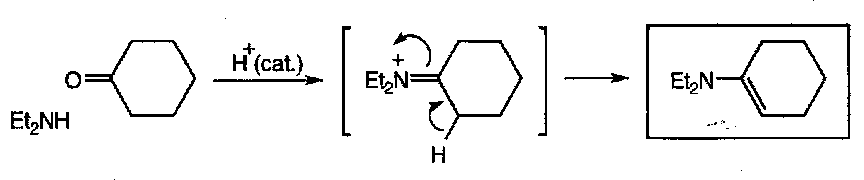
Enamines
Using a secondary amine to attack a ketone instead of a primary amine often leads to the formation of enamines instead of imines, like so:
This occurs because of the lack of the second proton on the nitrogen that can be lost.
They almost always react as β-carbon nucleophiles (not at nitrogen), like enols. They are more reactive than enols due to the positive charge, but less reactive than negatively charged enolates.
Formation of Enamines
The formation is acid catalysed (slow otherwise), and requires the removal of water (prevents hydrolysis, as for imines).
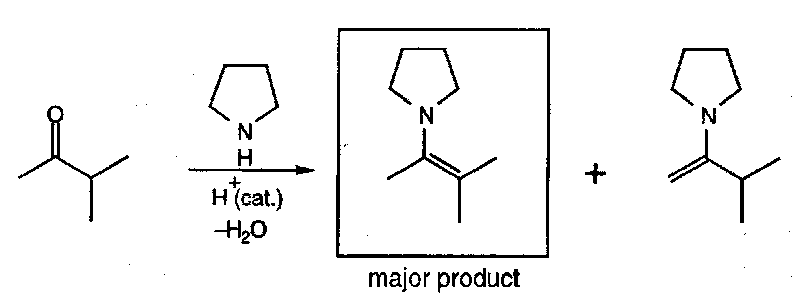
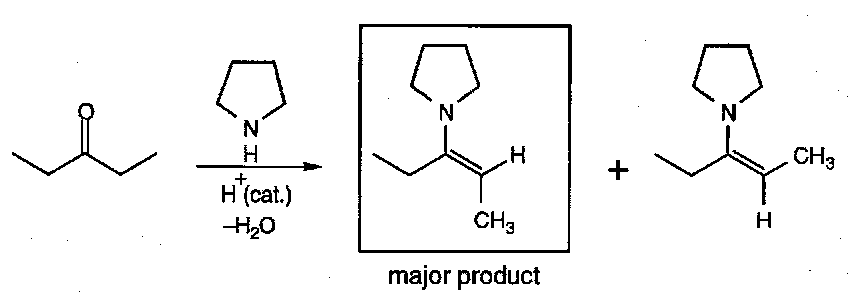
Unsymmetrical ketones can form two possible regioisomers. In practice, the more substituted double bond is strongly favoured:
In general, enamines are formed under reversible conditions (thermodynamic control), and the most stable alkene results (least sterically crowded):
Reactions of Enamines with Electrophiles
They react with alkyl halides, aldehydes, ketones, conjugated ketones and acid chlorides, i.e. anything normally attacked by enols/enolates.
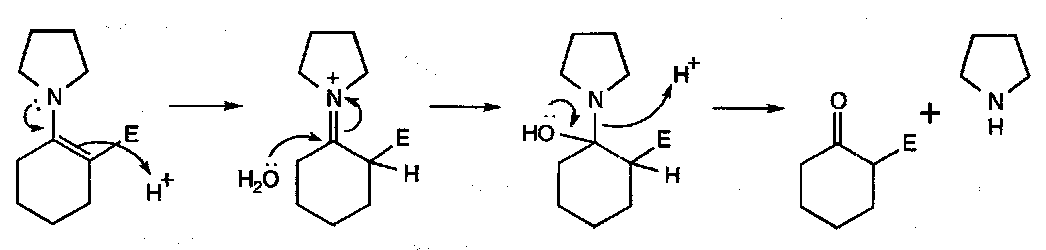
Hydrolysis of Enamines
This proceeds via the imine:
Enamine chemistry is often not used for substitution at the β-carbon, however. This is because of the recent advances in enolate chemistry that allow greater control over the products. Enamine chemistry is widely used for initiating intramolecular reactions, however:
Amides
- Planar.
- Relatively inert.
- Non-basic.
- Weakly nucleophilic at oxygen.
- H-bond acceptor and donor.
Synthesis of Amides
Condensation between an activated carboxyl acid derivative and an amine (see Peptides Notes). The former is usually an acid chloride or acid anhydride for simple amide bonds.
Note of course the use of one-pot coupling reagents:
Carbodiimides:
Acyl Azides:
Reactions of Amides
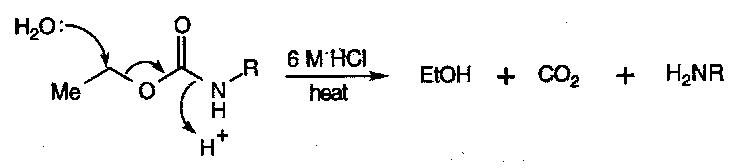
Hydrolysis – requires very forcing conditions (strong acid or base).
Hydrazinolysis – powerful nucleophiles like hydrazine can react with amides, displacing H2NR.
Vilsmeyer Reaction –
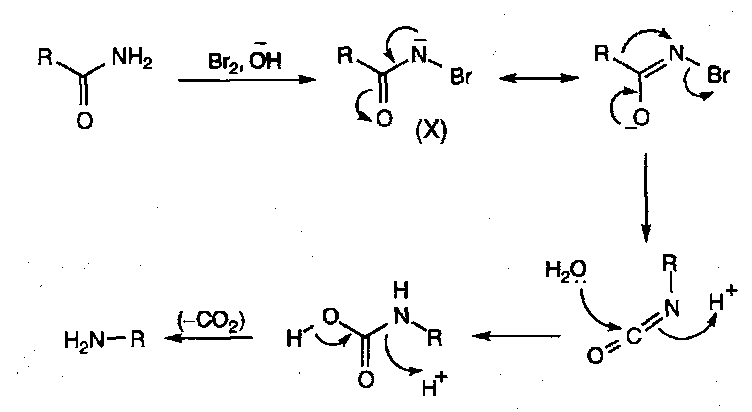
Hoffmann Rearrangement –
Loss of “CO” from the amide bond, making an amine.
Nitriles
-CN is a nucleophile, but not very basic at all.
Formation of nitriles
SN2 reactions using inorganic –CN.
Attacking carbonyls can be fruitless, as the equilibrium favours the carbonyl unless acid is added, but H+ reacts with –CN and slows the reaction. HCN can be used, which gives rise to the cyanohydrin R1R2C(OH)(CN).
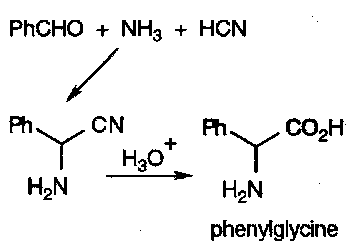
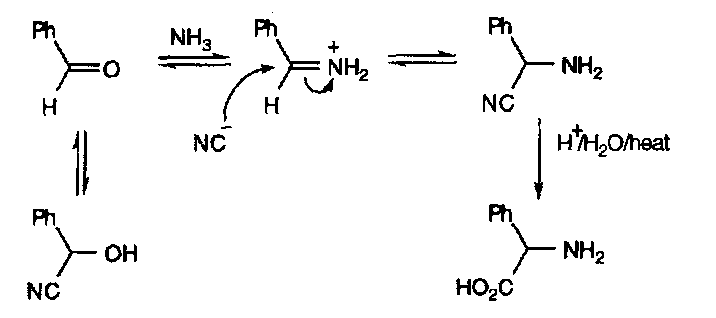
This equilibrium is exploited in the Strecker Synthesis of α-amino acids –
The pH for this reaction is critical (pH=7). Too acidic, and there is no free NH3, while too basic and the imine won’t react. KCN, NH4Cl and NH3(aq) was found to naturally buffered to about the right pH. The strength of the C-N bond drives the equilibria to the right.
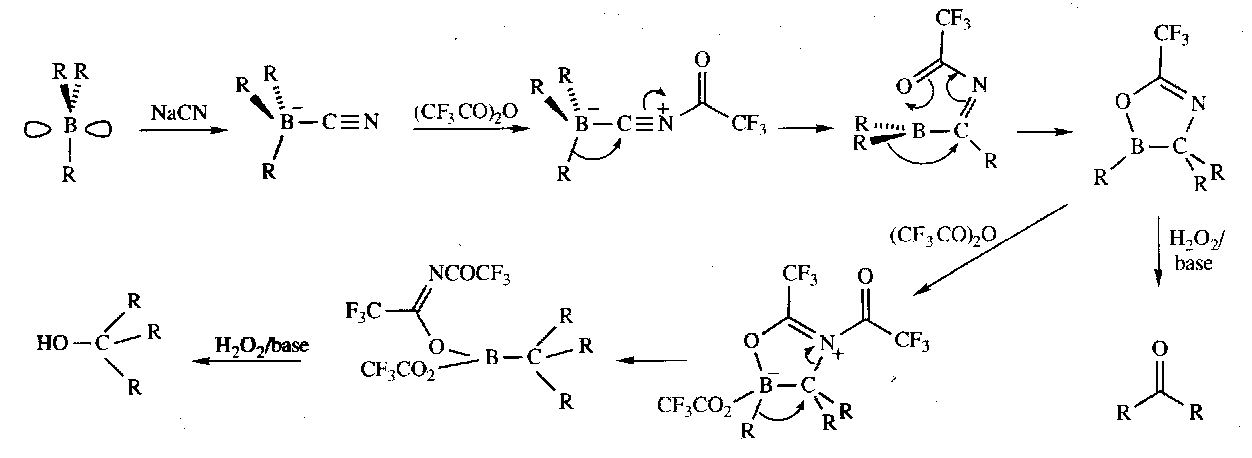
Nitriles from primary amides
Occurs by loss of water, but requires harsh conditions, such as PCl5, carbodiimide, or (CF3CO)2O. In all cases, the mechanism involves attaching a powerful electrophile to oxygen, and making it a good leaving group.
Reactions of Nitriles
Hydrolysis –
Can obtain carboxylic acids or carboxylates by using strong acid or base. This also allows esters to be formed in a single step, by adding the appropriate alcohol.
Nucleophilic Attack –
Powerful nucleophiles can attack the C. Helps if the N can be protonated with strong acid, or Lewis Acid (e.g. reactions with Grignard Reagents).
Enolate-type Chemistry –
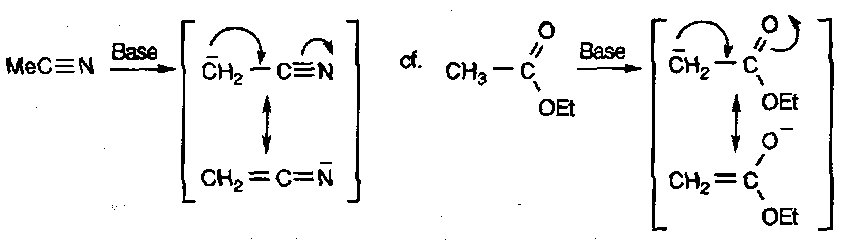
With powerful bases, nitriles can be deprotonated, to give the nitrogen equivalent of an ester enolate.
As expected, reaction usually occurs at the C rather than the N.
It should also be noted that nitriles offer excellent additional stability to traditional enolates (or other delocalised anions).
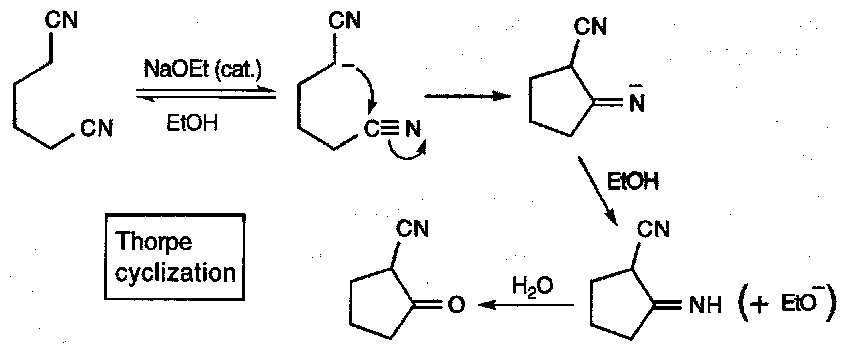
Nitrile “enolates” can be effectively trapped and cyclised when the chain is long enough, e.g. the Thorpe Reaction:
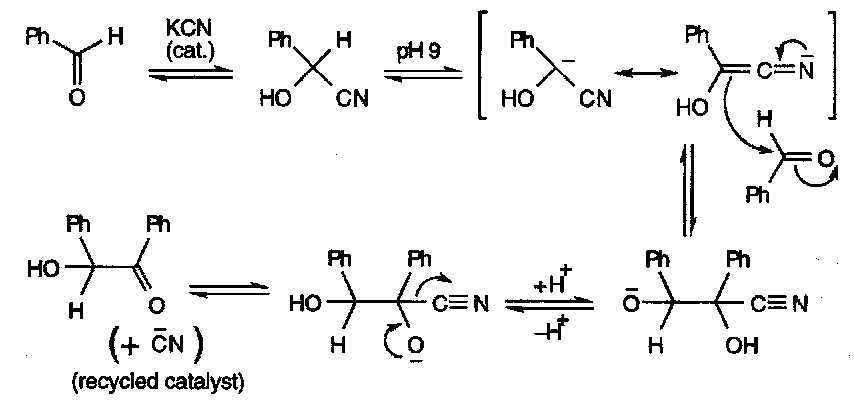
Nitrile deprotonation is also seen in the Benzoin Condensation:
Reduction –
Reduction of nitriles can be achieved by only strong reducing agents, such as LiAlH4 and DIBAL (the latter stops at the aldehyde).
Urethanes, Ureas, Imides and Diimides
These are all of the type R-X-Y-Z-R’.
Urethanes
These are of enormous importance as readily removable N-protecting groups:
(See Peptide Notes for more on these).
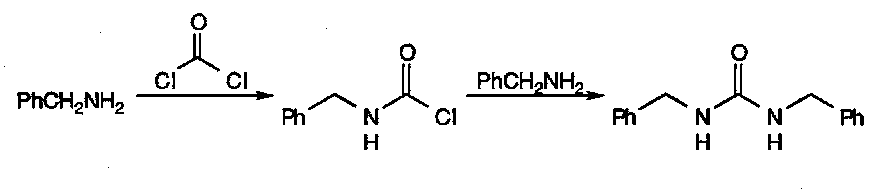
Ureas
Synthesised from two equivalents of amine with one equivalent of phosgene:
They are generally quite unreactive themselves, as the carbonyl is stabilised by two +M nitrogens. Strong alkaline hydrolysis yields amines + CO2.
The only other useful reaction is dehydration, which requires very powerful reagents, but generates valuable diimides:
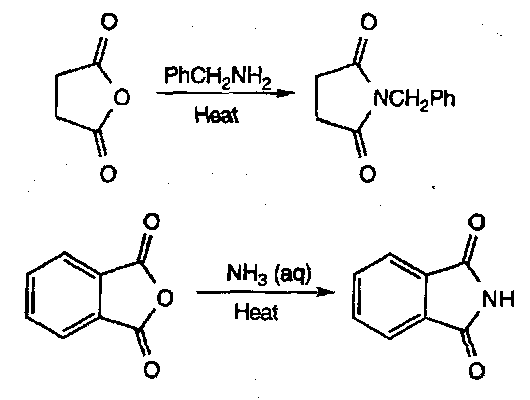
Imides
These are synthesised by heating cyclic anhydrides with ammonia or a primary amine:
The mechanism is:
There are two main types of reaction.
The first is when the imide has an NH group, which is deprotonated to give a stabilised anion. The nucleophilic nitrogen can then be monoalkylated readily. This is important as the alkylation can only occur once (as in the Gabriel Synthesis).
Imides with an N-R group react quite efficiently, and again once only. For example:
Diimides
These are synthesised by dehydrating ureas (see above).
They are superb reagents for achieving one particular type of reaction: turning carboxylic acids into esters or amides.
DCC is one of the most widely used reagents for preparing peptides (see above).
(Much more on this in Peptides Notes).
Compounds with N-N Bonds
- Hydrazine – RNH-NH2.
- Hydrazide – RCONHNH2.
- Hydrazone – RCH=N-NH2.
- Diazo – R-N=N-R.
- Azide – R-N=N+=N-.
Hydrazines and Hydrazides
Generally reactions revolve around the formation of hydrazones with aldehydes and ketones. This is due to the nucleophilic NH2 component, which forms an imine.
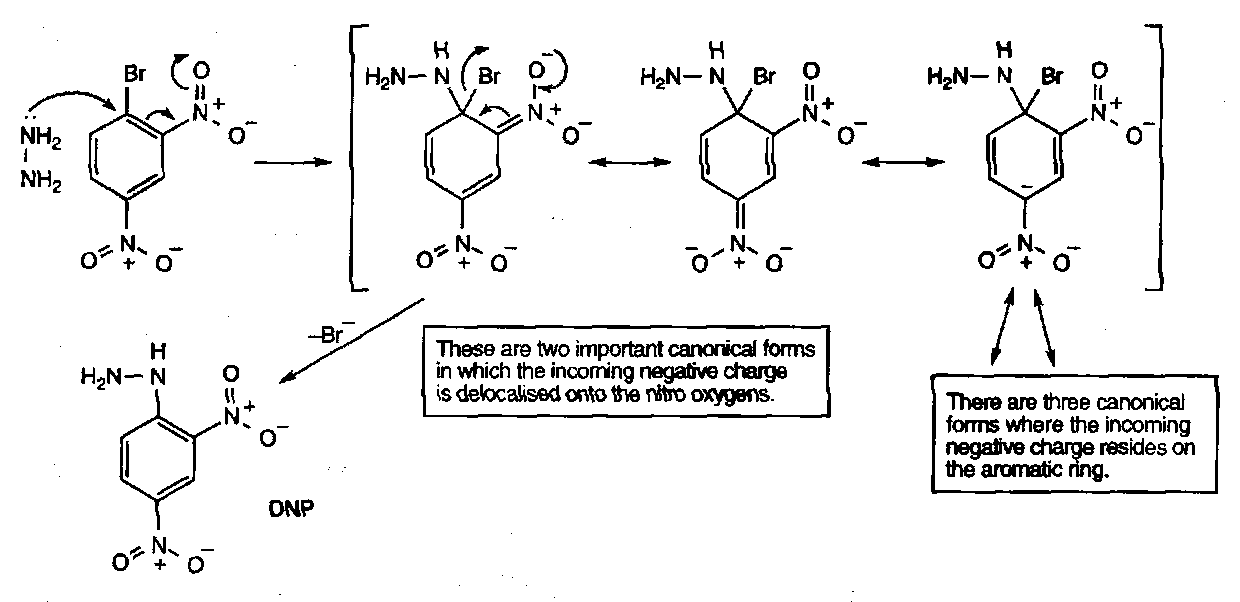
2,4-dinitrophenylhydrazine (DNP) is widely used and gives a bright orange precipitate, and is often used as a test for ketones / aldehydes (before the advent of spectroscopy).
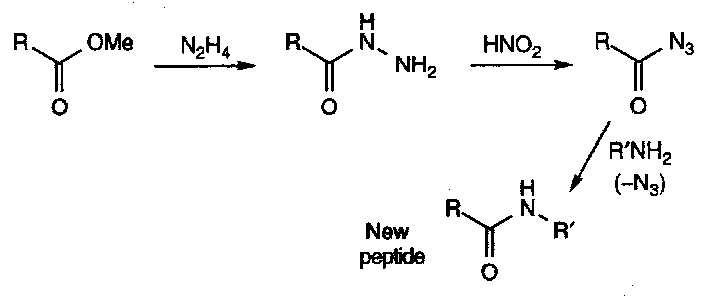
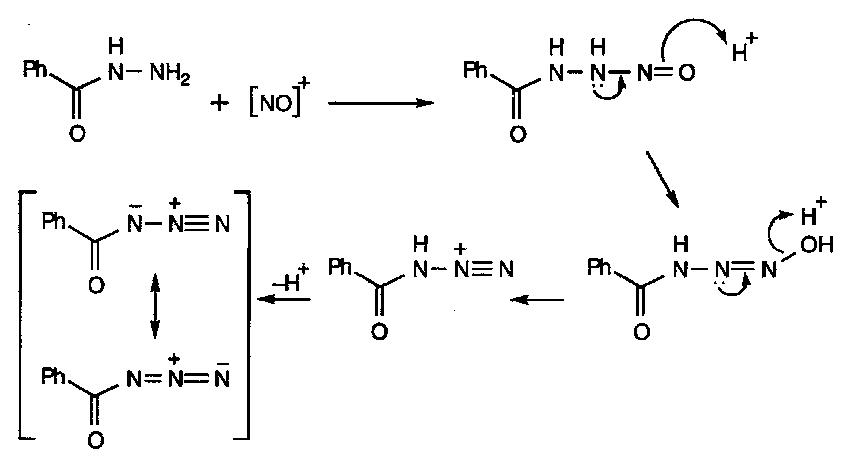
Acyl hydrazines (hydrazides) have strongly differentiated nitrogens, which can be exploited in reaction with nitrous acid, which forms the acyl azide:
2HNO2 ⇌ [NO]+ + H2O + NO2-
Acyl azides are useful acylating agents, particularly in peptide synthesis.
Hydrazones
Formed from hydrazine + carbonyl with loss of water. They are much less reactive than imines (requires very strong acid to regenerate the ketone).
An important reaction of them is the Wolff-Kischner Reduction –
Diazo Compounds
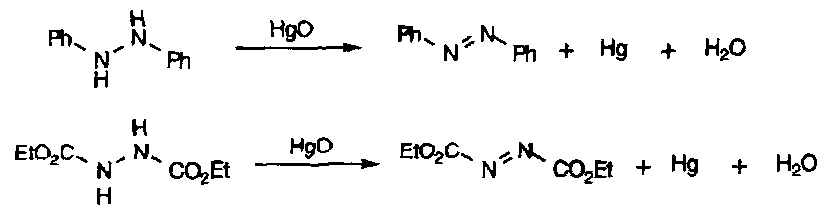
These are trickier to prepare – requires the groups on either side of the N-N bond to be fully substituted to prevent hydrazone formation. Thus, oxidation of certain hydrazines leads to their formation:
Diazo dyes are prepared by trapping out the diazonium compounds with a carbon nucleophile:
They undergo few useful reactions, and are mainly of importance in the dye industry.
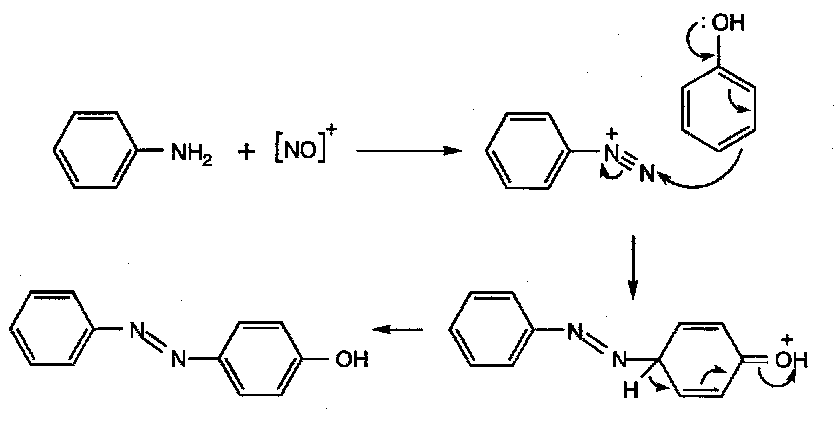
Aromatic diazonium compounds are useful in aromatic synthesis, by the Sandmeyer Reaction –
Aliphatic diazonium compounds usually collapse to alcohols almost straight away, releasing N2.
Azides
These are prepared by nucleophilic attack of NaN3 on a suitable alkyl compound such as a halide, or an acyl chloride for acyl azides. The azide is mononucleophilic, as has already been mentioned.
They are often used as intermediates in the synthesis of primary amines, and acyl azides find use in peptide synthesis (activated carboxyl).
Irradiation or heating of azides gives highly reactive nitrenes (See Reactive Intermediates Notes).
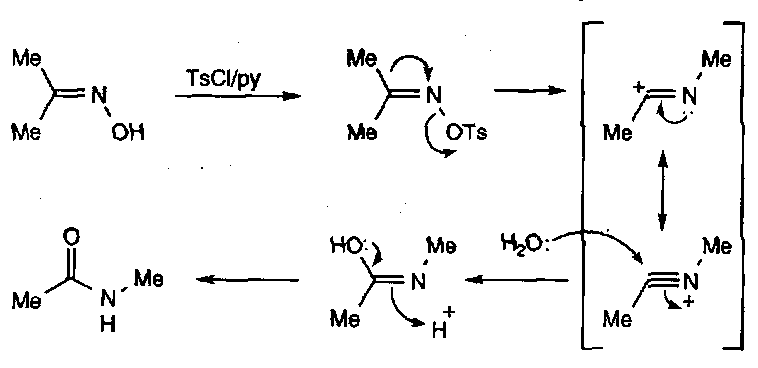
Oximes
Oxime – R-CH=N-OH
Note the stereochemistry – the lone pair means there are E and Z forms.
Synthesised from carbonyl + hydroxylamine (H2N-OH).
Reactions of Oximes
Hydrolysis – they are relatively inert due to lone pair conjugation, but vigorous conditions will cleave the C=N unit to regenerate the carbonyl.
Reduction – hydrogenation over highly active catalysts leads to reduction of the π-bond and the N-O σ-bond, giving primary amines.
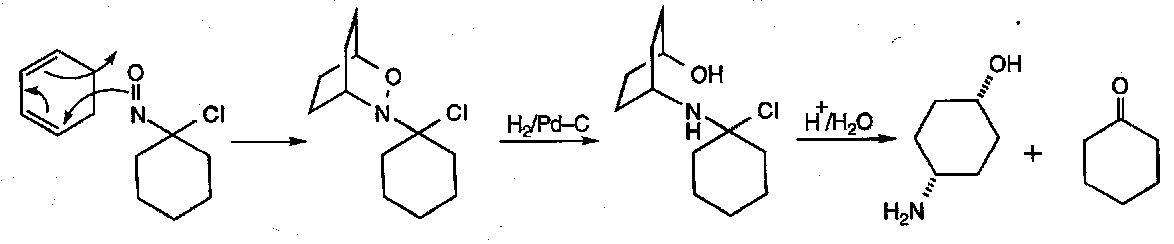
Dehydration – with oximes derived from aldehydes, H2O can be eliminated to generate the nitrile. Powerful dehydrating agents are required, and the leaving group nature of OH must be improved. PCl5 and H2SO4 (conc.) are suitable reagents. Yields are often poor though due to competing Beckmann Rearrangement.
Beckmann Rearrangement
Oximes of ketones readily undergo this reaction.
If an unsymmetrical oximes is used, only the alkyl/aryl group anti to the OH will migrate. This means that E and Z isomers give different products.
Note that oximes derived from aldehydes have competing dehydration occurring.
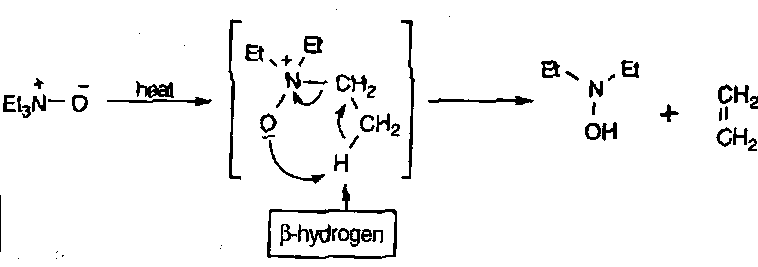
N-Oxides
N-Oxide – R3N+-O-.
Unusually, the N-heteroatom bond of N-oxides is not introduced already intact, but is delivered by oxidation of N. Peroxides or Peracids are usually good for this procedure.
Reactions
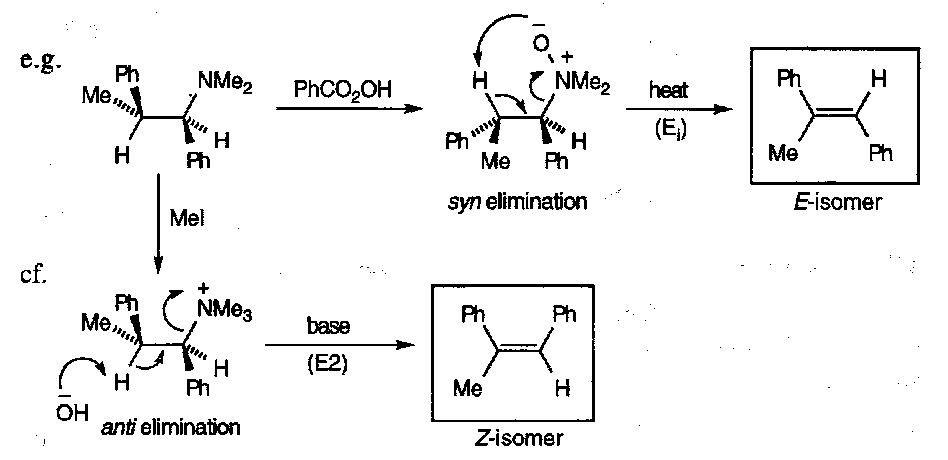
β-hydrogen SYN elimination can occur, when the β-H is present and with heat:
Hence, it can be possible to form either E or Z isomers by changing reaction conditions, e.g. peracids vs. base:
Pyridine N-Oxide is useful as it makes pyridine far more reactive (in fact completely changing its reactivity). This allows it to be nitrated (and other similar aromatic reactions are suitable). The N-O bond can then be cleaved afterwards by treatment with PCl3.
Nitroso Compounds
Nitroso – R-N=O and N-Nitroso – R2N-N=O.
C-Nitroso compounds are tautomeric with oximes, but oximes are thermodynamically more stable.
Aliphatic C-nitroso compounds
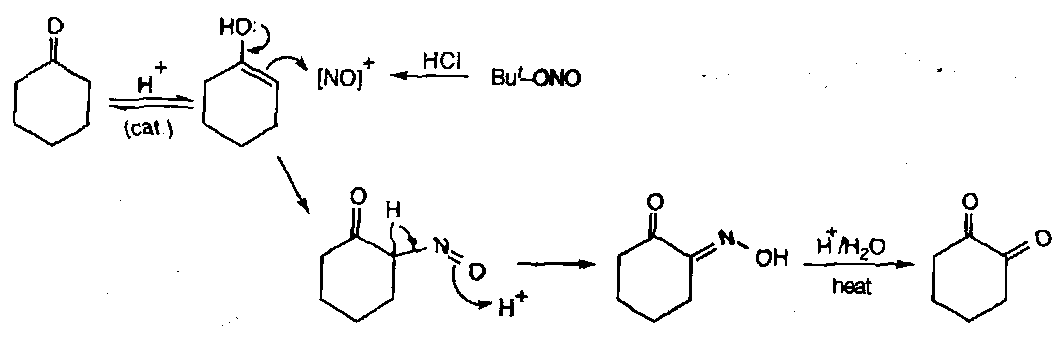
Difficult to synthesise due to trapping [NO]+ with a nucleophile leading to further reactions. Usual method is to treat a ketone with NOCl:
This compound can actually function as a dienophile in Diels-Alder reactions, which makes it a valuable route to 4-hydroxyamines:
Similar reactions can be performed with acyl nitroso compounds (made from acyl chloride + H2N-OH + IO4-).
Aromatic C-nitroso compounds
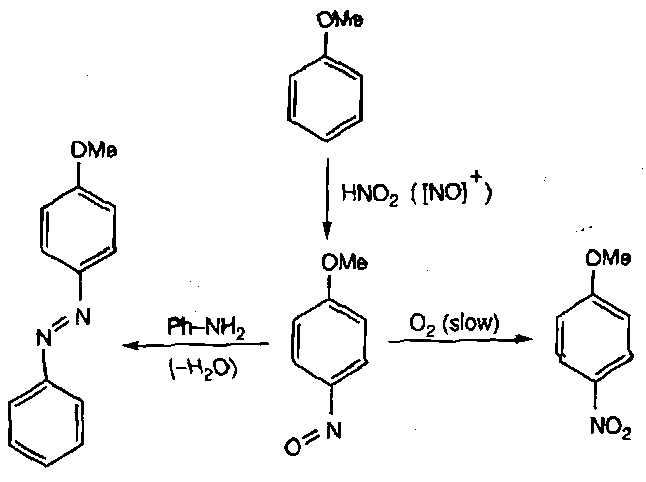
These cannot tautomerise to an oxime, and are further stabilised by conjugation. Thus, nitrous acid can be used in synthesising them from activated aromatic compounds. They have few useful reactions though.
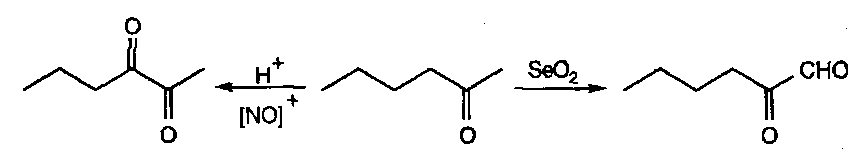
α-diketones via nitroso compounds
This exploits the tautomerisation, and utilises the enol tautomer of the ketone as a carbon nucleophile that picks up NO+:
Acid catalyses the enolisation, the NO+ formation, and the hydrolysis of the oxime!
This reaction is useful for reverse regiocontrol to the SeO2 dioxide oxidation of a ketone:
N-nitroso compounds
These can distinguish primary, secondary and tertiary amines. By treating them with nitrous acid, NO+ is trapped by the nitrogen nucleophile, except for tertiary (no reaction). The primary amines lose water rapidly though, generating the aliphatic diazonium ion which collapses. This leaves secondary amines forming N-nitroso compounds which are usually orange oils.
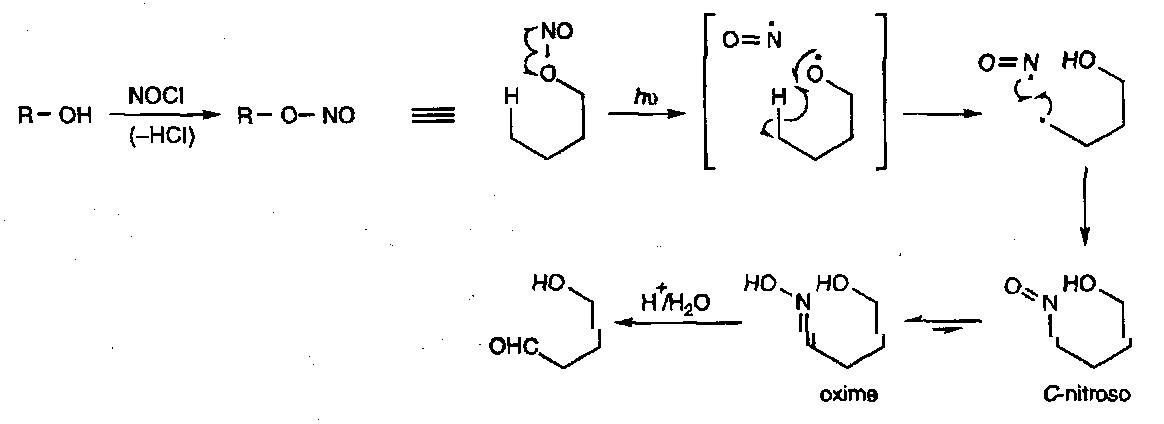
O-nitroso compounds
These are also known as nitrite esters, and undergo the important Barton Reaction:
The reaction is important as it allows the functionalisation of remote positions by H• abstraction.
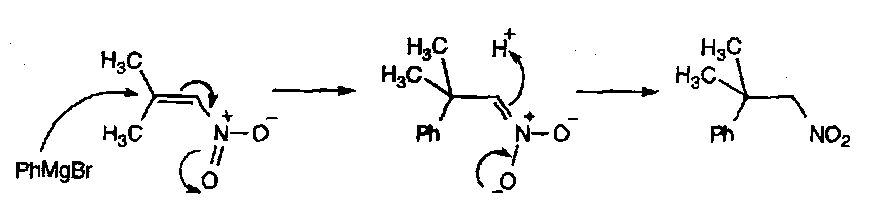
Nitro Compounds
Nitro – R-NO2.
Aromatic Nitro Compounds
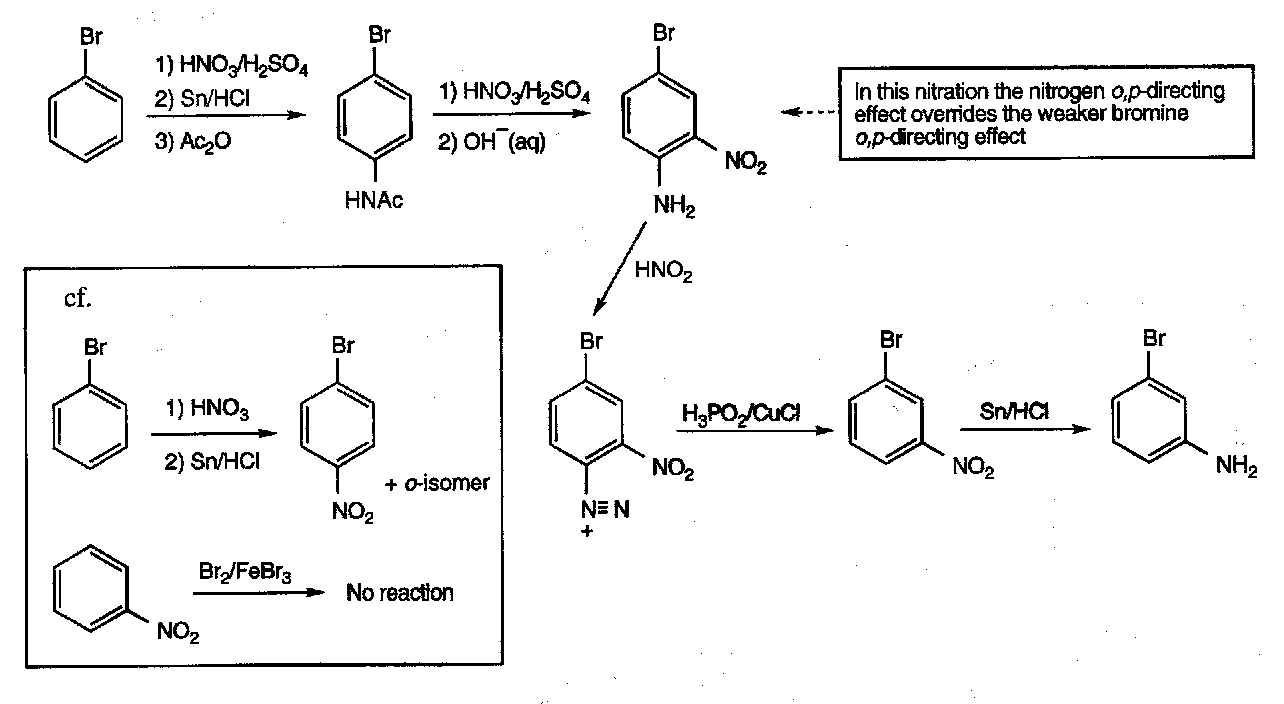
Synthesised by direct nitration of aromatic compounds.
2HNO3 + H2SO4 ⇌ H3O+ + NO3- + HSO4- + [NO2]+
Nitration generates a strongly deactivated product, so further nitrations require very forcing conditions.
They can be useful in forming aromatic amines. This is effected by reduction, typically using Sn/HCl, H2/Pd-C, or LiAlH4.
Completely removing the nitro group requires reduction to the amine, then diazo formation (with HNO2), and then reductive loss of N2 (achieved by adding NaBH4 or hypophosphorus acid/Cu+ mixture). This is often done when the strongly directing effects of the amine group are needed (but not required in the product).
Nitro groups can also allow unusual reactions on the aromatic ring, e.g.
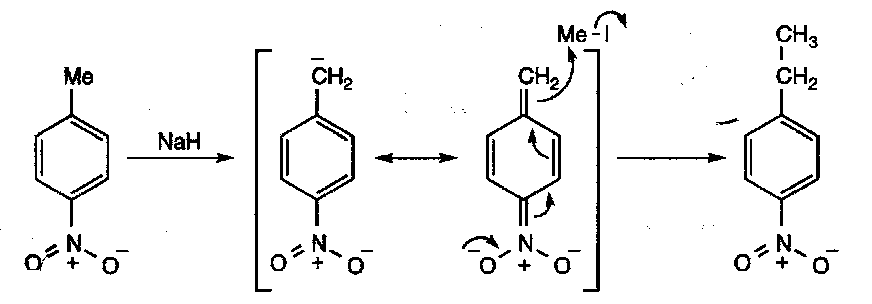
Similarly, alkyl groups ortho or para to a nitro group can be deprotonated:
Aliphatic Nitro Compounds
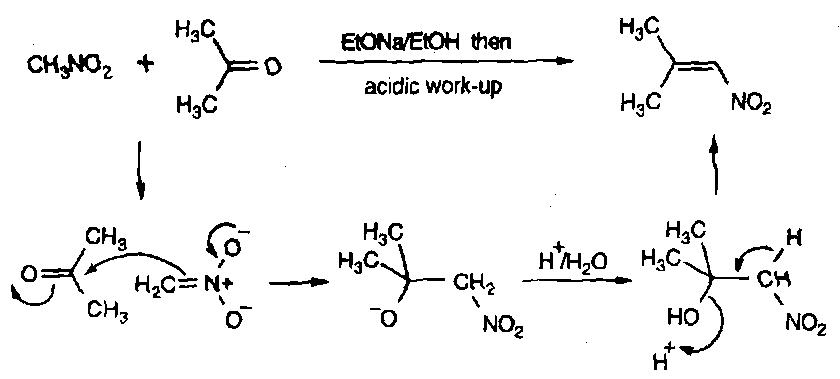
Nitroalkanes are very useful in “enolate” type chemistry. They are highly acidic and analogous to carbonyls as a result.
The advantage with them is they have little propensity to self-condense (reversible attack at N+ does not generate stable products), so they can take part in cross-condensation reactions effectively.
Similarly, they can be attacked in a Michael Addition type reaction:
It is useful, after this chemistry has been carried out, to be able to convert the nitro group into something else. There are two highly efficient methods to turn it into a ketone or amine.
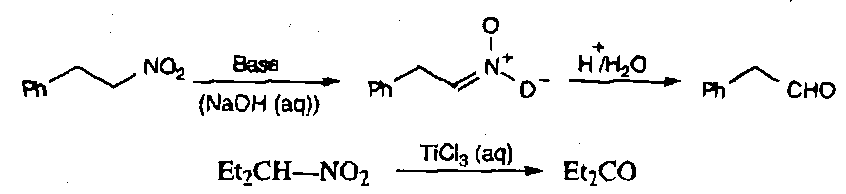
Hydrolysis – requires vigorous conditions, and is called the Nef Reaction. Also requires an α proton to be present.
Reductive hydrolysis by TiCl3(aq) is usually more efficient, and involves one electron reductions.
Reduction – many reducing agents will convert nitro groups into amines. Usually use LiAlH4 or H2/Pd-C.
BORON
Boron often forms compounds that are Lewis Acids, as in BBr3, where it will complex by accepting electrons, often from O.
Hydroboration
Adding B and H to a double bond.
Boron adds preferentially to least hindered end of C=C (anti-Markovnikov result).
However, using BH3 will lead to a tri-substituted boron. Alkylboranes are commonly used instead, e.g. 9-BBN or thexylborane:
These react via mechanisms often governed by sterics, and also show a much greater degree of stereoselectivity.
Organoborane compounds that form from these reactions are easily oxidised. The common reagent used is hydrogen peroxide in alkali.
Water / alkali then converts this to 3ROH.
Note that chromic acid (H2CrO4) can also be used to oxidise – this leads to the ketone as opposed to the alcohol.
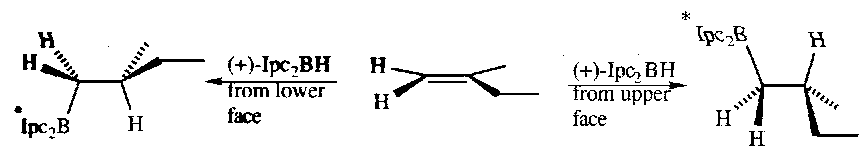
Asymmetric Hydroboration
Generating the hydroborating agents from homochiral (enantiomerically pure) alkylboranes allows asymmetric hydroboration reactions.
For example:
The efficiency of asymmetric hydroboration is high if one approach trajectory leads to severe steric interactions between the hydroborating reagent and the alkene and the approach trajectory on the other face of the alkene involves relatively insignificant interactions, i.e. the energy difference between the two transition states is large.
This is a common route to optical active alcohols.
Protonolysis
Abstracts a proton using alkylborane.
Halogenolysis
Organoborane after accepting a base becomes negatively charged. Thus the alkyl substituents can accept as nucleophiles under certain conditions:
Amination
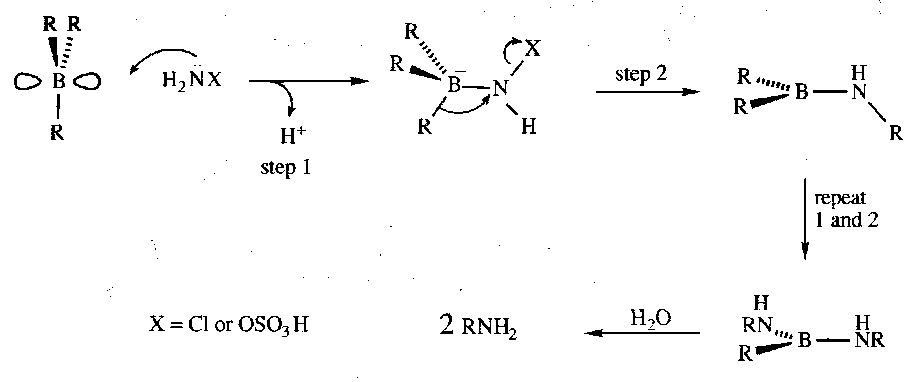
Alkylboranes are converted to primary amines by amines bearing good leaving groups, such as chloroamine or O-hydroxylaminesulphonic acid:
The reaction proceeds with retention of stereochemistry via the mechanism below:
i.e. cis addition of ammonia across a C=C bond.
Isomerisation
Heating alkylboranes causes boron to move to a position where steric interactions are minimised:
Carbonylation
Turn organoboranes into ketones and alcohols by reaction with CO.
2nd migrations need H2O or ROH present, while the 3rd migration needs HO-CH2-CH2-OH.
A similar reaction with the cyanide anion (isoelectronic with CO) can yield alcohols and ketones too:
It is experimentally simpler and occurs under milder conditions that Carbonylation.
Alkene Synthesis
Can produce both E- and Z-alkenes using organoboranes.
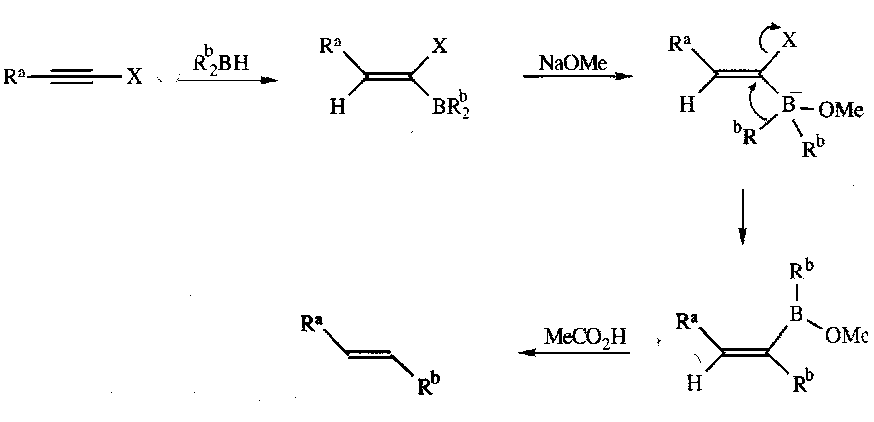
E-alkenes
Hydroboration of 1-halo-alk-1-ynes followed by treatment with NaOMe and then acetic acid:
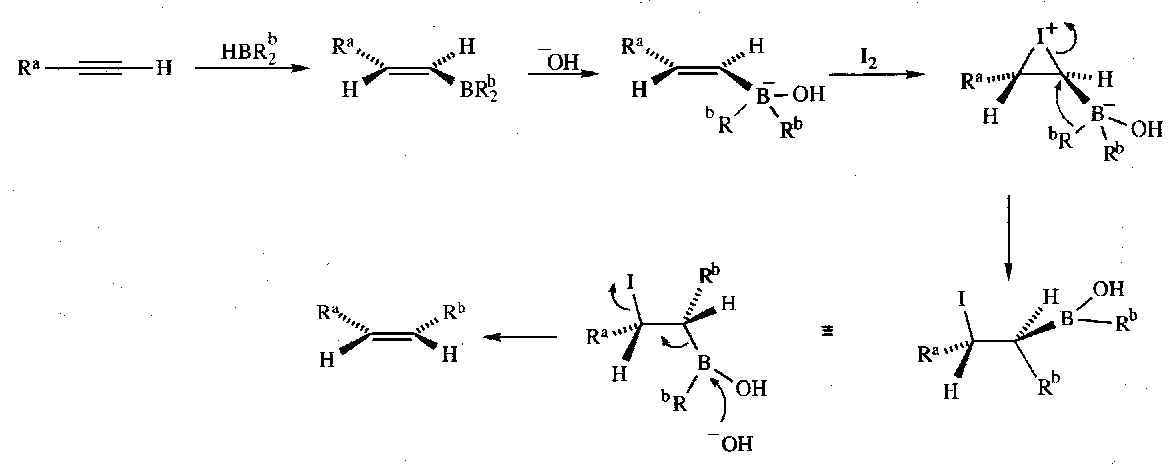
Z-alkenes
Hydroboration of alk-1-ynes followed by addition of NaOH and iodine gives Z-alkenes:
The iodonium species formed creates an electrophilic terminus for alkyl migration, which occurs with retention of configuration of the alkyl group. Antiperiplanar elimination then gives the Z product.
Allylboranes
Allylboranes also react well with ketones. This occurs as:
The transition state for the first stage of the mechanism takes the chair form. This is useful, as it allows prediction of the stereochemistry of the product, since the bulky R will always be equatorial. The result as shown is 3 lots of homoallylic alcohol.
It is worth noting that at room temperature they rearrange, and the stereochemistry of the allylborane is scrambled. Thus, they are typically used at low temperature if one predominant isomer is needed. Also, OR/NR2 groups π-donate into vacant p orbitals and suppress the rearrangement.
Boron Enolates
In a similar way to allylboranes, these reagents are useful as they proceed via a chair transition state (allows stereochemical control).
Can also obtain kinetic / thermodynamic enolates as required (as for other types of enolate):
Reduction
Boron reagents are often highly useful in reduction reactions. Typical reagents are borane itself, and also borohydrides. More about this can be found in the Oxidation and Reduction Notes.
SILICON
Organosilicon chemistry is typically drive by the strong SiO and SiF bonds.
Reactions often occur via an SN2-Si
pathway, which involves addition-elimination via an ate complex. It is extremely
rapid.
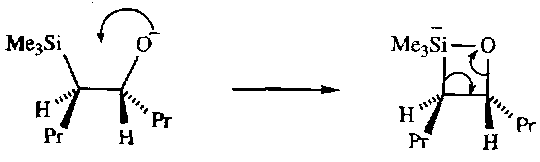
Formation of Carbocations on C atom β to Si is also common, i.e. Si-C-C+. This is stabilised due to overlap between the π on Cβ and σ on Si. This requires that the orbitals are in the same plane, so it is often not found to occur in cyclic systems.
Formation of Si-C- also occurs. Stabilisation now occurs because of d-orbital overlap and also overlap with σ* on an adjacent C-Si bond.
Trimethylsilane
This is typically a protecting group for hydroxyl. It can utilised by adding TMSCl to R-OH in a weak base. Other substituents besides methyl can be used, and the Cl can be swapped for e.g. I or pyrrole to make the protecting group formation reaction very rapid.
Modifications can also be made to resist cleavage by acid / base, hydrogenolysis or attack by nucleophiles. This can be done by reducing lability, e.g. TMSiOR is very labile, but tBuMe2SiOR or tBuPh2SiOR are much less so.
Cleavage of the protecting group is best achieved by using F- as Bu4NF. Strong OH/H+ or nucleophiles will also do this, however.
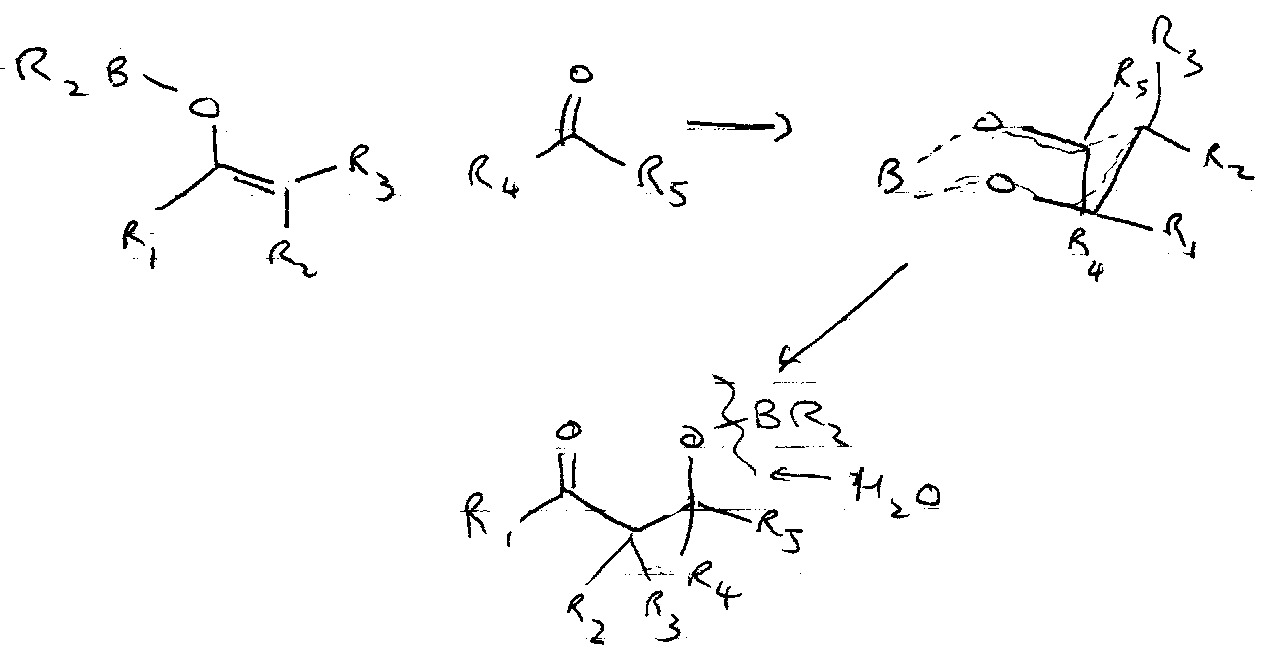
Silyl Enol Ethers
These are formed by trapping normal enolates, and can be made more reactive, e.g.
They also react with strong electrophiles:
It is particularly useful to trap the kinetic or thermodynamic enolates out using these. Under kinetic conditions, deprotonation occurs at the least substituted carbon, so the enolate anion with the least substituted double bond forms in excess.
Under thermodynamic conditions, the equilibrium between the two anions favours the more substituted C=C.
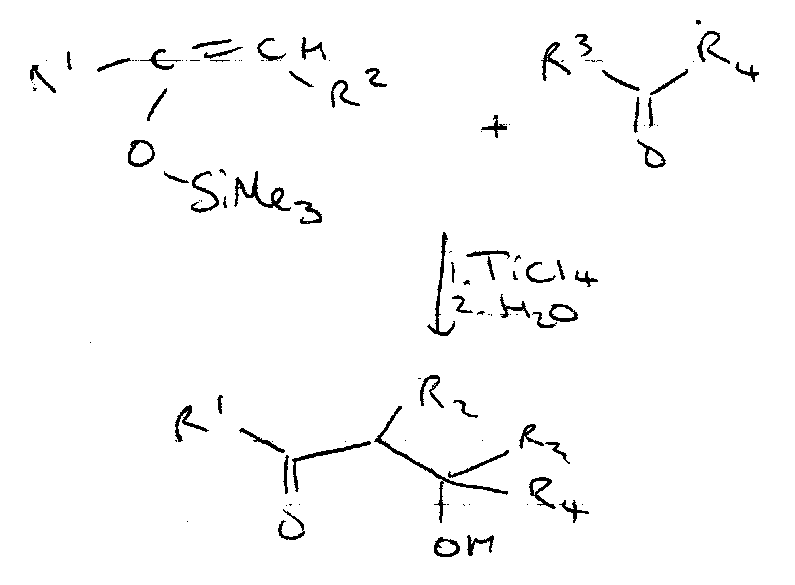
Mukiyama Reaction
Alkene Synthesis (Peterson)
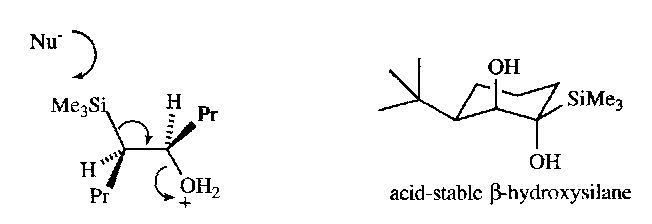
These methods form β-hydroxysilanes. These are then converted into alkenes by elimination. Can use either an acid (ANTI) or a base (SYN) pathway:
Acidic Conditions –
Basic Conditions –
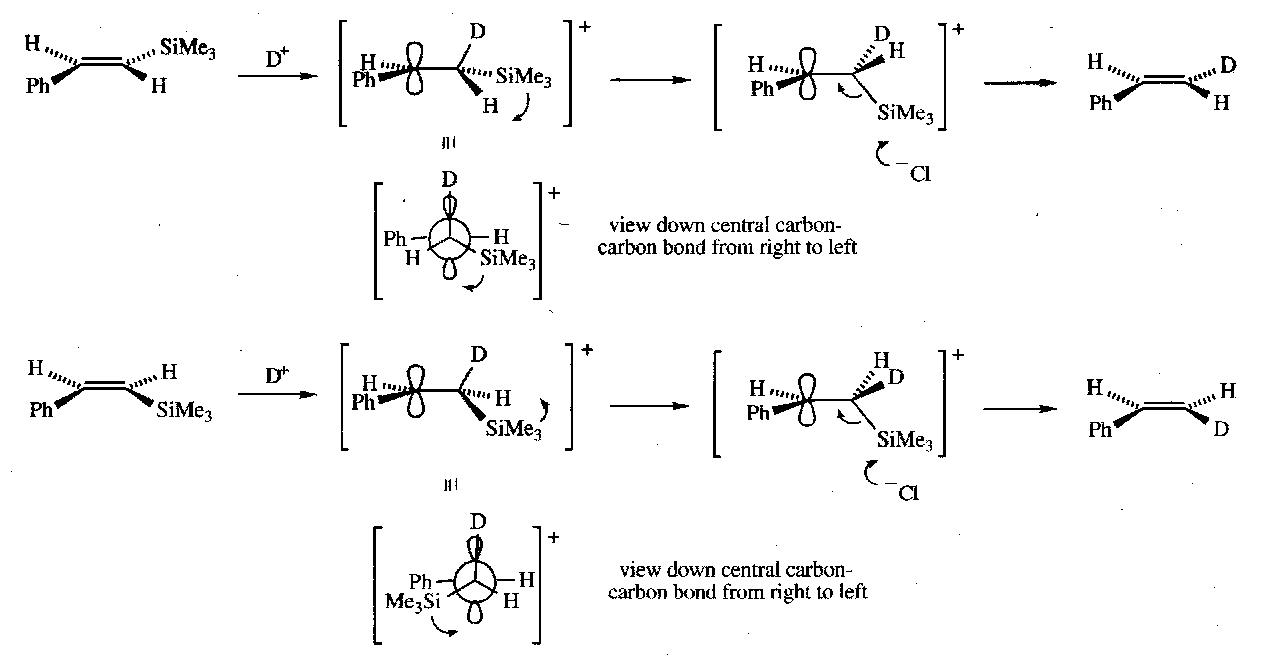
Vinyl Silanes
Can be made by hydrosilylation (e.g. with Et3SiH), partial hydrogenation of alkynylsilanes, or replacement of the halogen in a vinyl halide with a metal in conjunction with an electrophilic quench (e.g. Mg + Me3SiCl).
They undergo electrophilic substitution reactions, e.g.
Nazarov Cyclisation
A particular example of an electrophilic substitution at a vinyl silane.
Vinyl silanes also undergo nucleophilic addition under basic conditions, as the carbanion α to the Si can be stabilised.
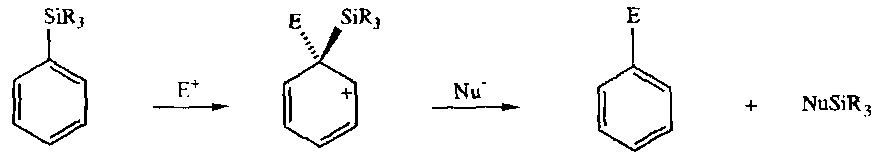
Aryl Silanes
Formed by deprotonation or metal-halogen exchange.
Their main use is ipso-desilylation. The overall result of this is that an incoming electrophile replaces the silyl substituent on the aryl ring.
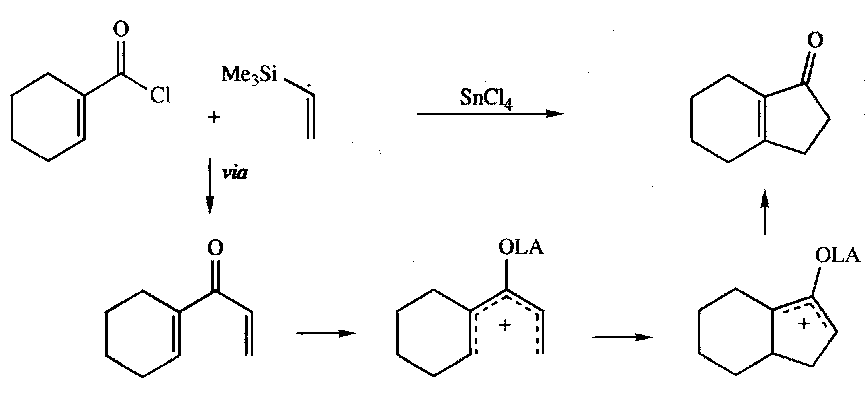
Allyl Silanes
Attack electrophiles from γ-carbon, due to stabilisation of the carbocation at β.
Sakurai Reaction
An example of their application is the Sakurai Reaction:
Can also use:
|
|
silyl enol ether |
|
|
silyl ketene acetal |
PHOSPHORUS
This is useful in organic chemistry for several reasons:
- Easily converts between III and V oxidation states.
- Nucleophilic
- P=O bonds are very strong (thermodynamically driven).
- Stabilises adjacent anions well.
When tervalent, it has a lone pair, so is a nucleophile. It is better than nitrogen, but consequently a weaker base, due to its increased size.
Corey Winter Desulphurisation
Phosphonium Salts
Arbusov
The product is broken down by reducing agents such as LiAlH4.
Perkov
Wittig
This turns carbonyls into alkenes, and works on a wide range of reagents.
A modification is the Wadsworth-Horner-Emmons Reaction. The phosphonate anion is more nucleophilic, so good for unreactive ketones. It also forms a more stabilised intermediate, which tends to favour the thermodynamic product (as opposed to Wittig which is kinetically driven).
Staudinger
Uses iminophosphoranes.
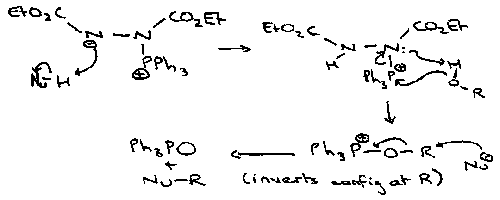
Mitsunobu Reaction
Uses DEAD – TPP:
Note that there is an inversion of configuration at R.
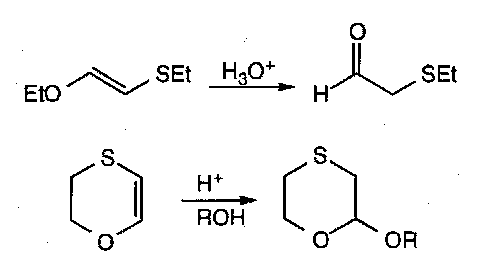
SULPHUR
Thiols
These take off alkyl groups from ethers:
Preparation is thus tricky, in order to avoid forming the sulphide. Protected sulphur derivatives that can only undergo one alkylation are used.
Chemistry of Thiols
High acidity (form salts). EtSH pKa = 10.5 (cf. ~16 for EtOH).
Promote nucleophilic substitutions at carbon.
Weak S-H bond.
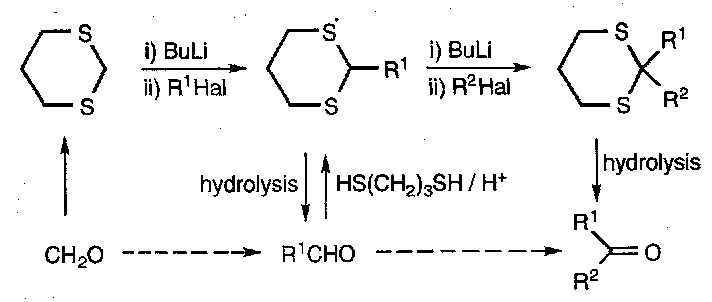
One use of their nucleophilicity is as protecting groups for aldehydes and ketones, as e.g. HS(CH2)2SH forms the dithioacetal by nucleophilic attack at the carbonyl. They can be removed by thiophilic reagents such as mercury, or Raney Nickel/H2 (the latter leads to the alkane from the original carbonyl).
One of the main features of dithioacetal chemistry is the metallation of simple formaldehyde and aldehyde dithioacetals. This comes about due to sulphur’s ability to stabilise the α carbanion.
Umpolung Chemistry is common with these (reversal of polarity):
Sulphides
Prepared by either nucleophilic attack on electrophilic carbon (as discussed above). Substitution with nucleophilic carbon attacking electrophilic sulphur can also be used:
Sulphides are oxidised by peroxides to sulphoxides and sulphones:
Peroxyacids can also be used.
Asymmetric oxidation can be affected by using the Sharpless method (diethyl tartrate, Ti(OiPr4), H2O / tBuOOH / -20oC).
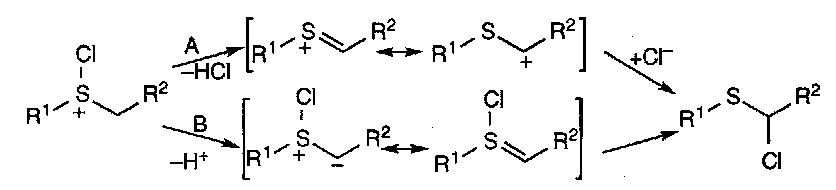
Suphides react with halogens to form halosulphonium salts. These intermediates are in equilibrium with tetracoordinate isomers, and easily hydrolyse to sulphoxides.
They are important in α-halogenation of sulphides:
These reactions are similar to the Pummerer Reaction (see below).
Vinyl Sulphides
Masked carbonyls (enol ether-like):
Nucleophilic addition of organometallic reagents can be affected at the α carbon.
Under strongly basic conditions vinyl sulphides possessing one or more γ hydrogens are interconverted with their allylic isomers by deprotonation-protonation, which leads to E/Z equilibration.
Sulphenic Acids
Simple sulphenic acids are unstable compounds, and sulphenyl halides are one of the more readily available of the dicoordinate sulphur compounds in this oxidation state.
Much of the chemistry of sulphenyl chlorides is dominated by their behaviour as reactive sulphenylating agents, e.g. on treatment with alcohols in the presence of base they are converted to sulphenate esters (R1SOR2)
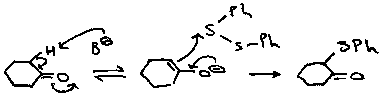
Sulphenic acids are principally electrophilic:
They are weak acids (similar to phenols) and exist in the RS.OH form rather than R.SO.H. Their most useful reaction is their addition to inactivated alkenes in a process which is the reverse of the pyrolytic elimination reaction of sulphoxides with a β-hydrogen (see later).
For example:
Sulphenate Esters
Allylic sulphenates are the most important class of sulphenates, because of the rapid equilibrium established with allylic sulphoxides via a [2,3] sigmatropic shift:
Disulphides
Can be considered as thiosulphenates.
Preparation:
They are sulphenating agents.
Sulphonium Salts
Lawesson’s Reagent
This can be used to convert amides or esters into the equivalent thioamides and thioesters (i.e. replace the C=O with a C=S bond).
It also acts on alcohols, converting them to thiols, and turns phosphate esters into alkenes.
The mechanism is thought to proceed in a similar manner to the Wittig Reaction, with intermediates of the type:
Xanthates
Ylids
Sulphur forms a wide range of ylids:
Sulphonium ylids are classified as either non-stabilised (e.g. Me2S+CH2-) or stabilised (e.g. Me2S+CH-COMe).
They have great synthetic usefulness. One particular example is shown above – forming epoxides from carbonyls.
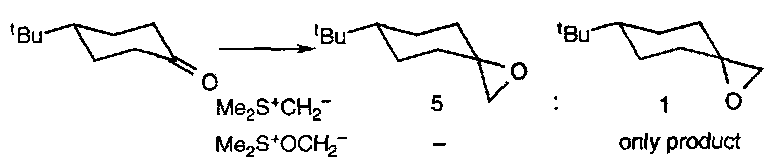
Stabilised and unstabilised ylids affect the stereochemical outcome of these reactions. For example, the epoxidation of 4-t-butylcyclohexanone proceeds as:
The unstabilised ylid predominantly gives epoxides derived from axial attack, while the more stable oxosulphonium leads to product from equatorial insertion only. It is likely that with the unstabilised ylid the addition step is effectively irreversible, and product stereochemistry reflects the inherent preference for axial attack on the unhindered carbonyl. Stabilised ylids have a reverse addition step, so product stereochemistry is determined by the more rapid cyclisations of the intermediate with the equatorial alkylsulphonium substituent.
Another characteristic difference between the two types of ylids is their attack on conjugated ketones. The stabilised ylid favours attack on the alkene (1,4), while the unstabilised ylid attacks the carbonyl (1,2) – soft vs. hard attack.
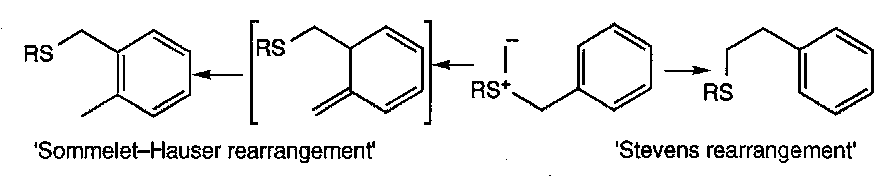
When benzylic sulphonium ylids are generated, it is found that two types of product can be formed depending on the particular substrate and the conditions used.
The one product type is obtained by a [2,3] sigmatropic shift onto the o-position of the aromatic ring followed by aromatisation (Sommelet-Hauser), while the other results from a 1,2 shift of the benzyl group (Stevens).
Sulphoxides
These undergo many types of reaction.
For example, they can be oxidised to sulphones by peroxides, or reduced to sulphides by phosphorus reagents.
Pyrolytic β-elimination:
Stereospecific SYN process, and can be used to introduce double bonds α to electron withdrawing groups or to isomerise a sulphoxides to another via elimination-addition, as shown.
As Nucleophiles
Sulphoxides with at least one α C-H bond are sufficiently acidic to undergo deprotonation on treatment with strong bases. The resulting α-sulphinyl carbanions are useful, e.g.
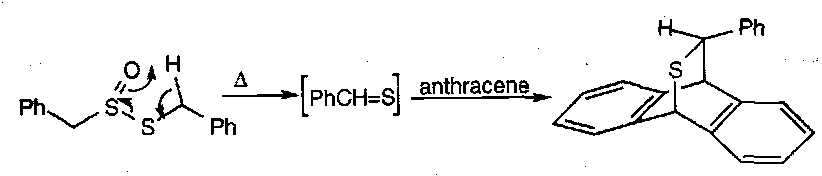
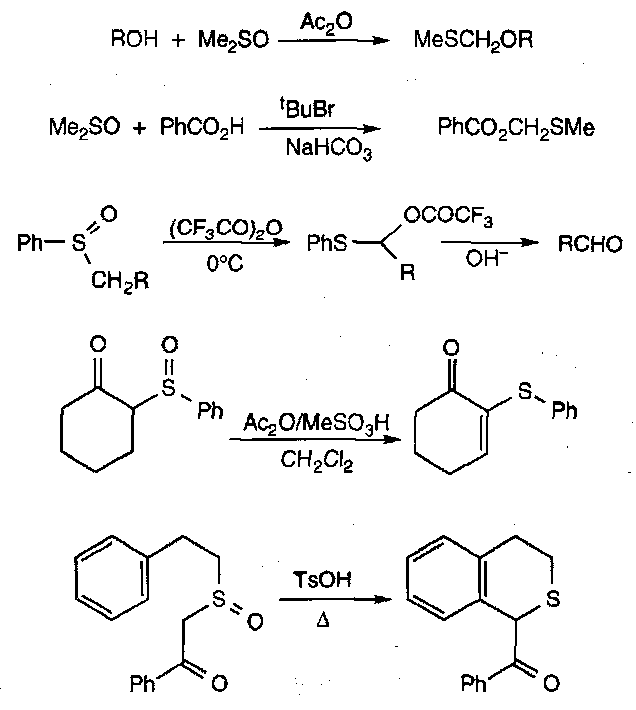
The Pummerer Reaction
A fairly generalised reaction for describing processes where sulphoxides have their S=O bond broken and a group containing O or other electronegative substituents is incorporated α to the sulphur. The overall result of the transformation is thus reduction at sulphur and oxidation at the α CH position.
A simple example:
The sulphonium ylid formed often leads to reaction at the α position that can more readily sustain a negative charge (when an unsymmetrical sulphoxide).
The scope and synthetic implications of the Pummerer Reaction is wide. Some examples:
It should be noted, however, that chirality transfer from S to C is not generally feasible as they racemise under acidic conditions.
Sulphonate Esters
Sulphones
The most important property of these is the formation of α-sulphonyl carbanions, and their subsequent reactions with a range of electrophiles.
Some examples:
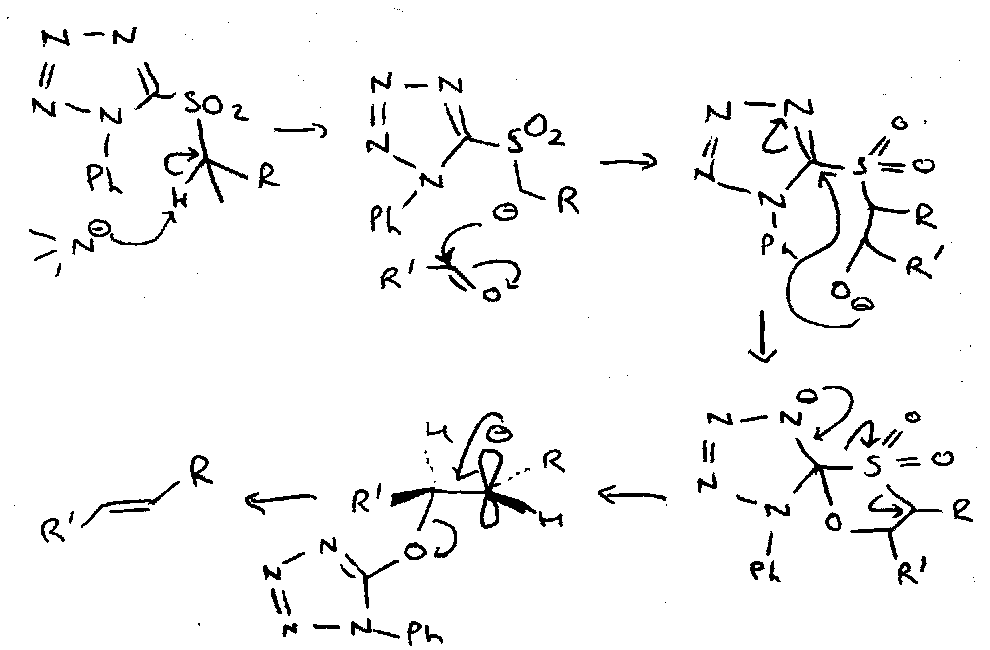
An important and characteristic reaction of these in compounds bearing a leaving group at the other α position is the Ramberg-Backlund Reaction, which gives rise to an alkene.
Reaction involves reversible α-sulphonyl carbanion formation, followed by intramolecular displacement of the leaving group to give an episulphones which looses SO2 by Chelotropic extrusion to leave the alkene. The product is derived by suprafacial loss of SO2, which is itself produced by inversion at both the α and α’ positions.
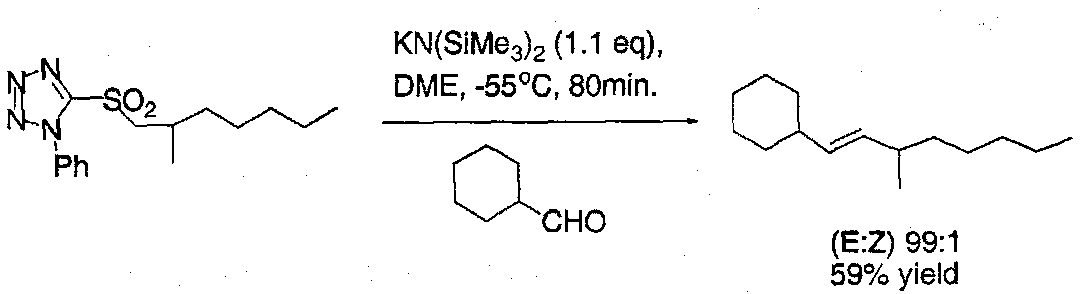
Julia Reaction
One of the best methods available for forming E-1,2-disubstituted alkenes from aldehydes in complex systems.
Kocienski’s Modification
Sulphonamides
α-sulphonamido carbanions can be formed under appropriate conditions.
Most reactions are based on sulphonylhydrazines, involving cleavage of the N-S bond with loss of a sulphinate ion:
An example is the Shapiro Reaction:
Also, Eschenmoser Fragmentation of α,β-epoxyketones:
SELENIUM
Compared to sulphur, it is larger and more polarisable, and has a lower electronegativity (same value as carbon). It forms weaker covalent bonds but shows a tendency to form more stable hypervalent species than sulphur.
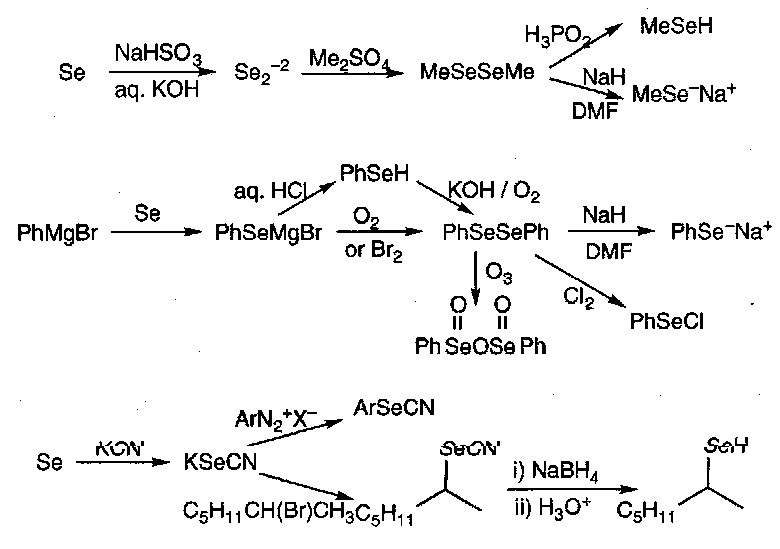
The chief starting materials for entry into organoselenium chemistry are selenols, diselenides and electrophilic selenylating agents (PhSeX).
Methods of Preparation:
Selenols are more acidic than thiols, and the larger Se atom makes a more powerful nucleophile (despite lower basicity compared to sulphur).
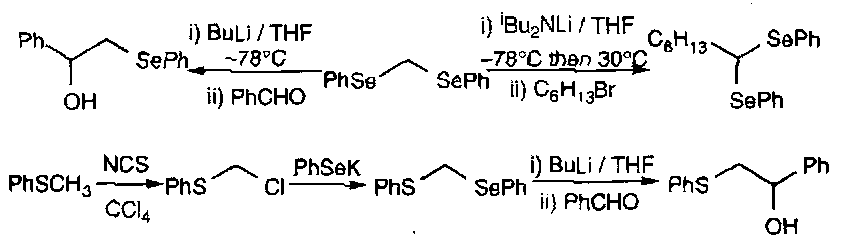
In a similar way to sulphur species, α carbanions can be stabilised, and are widely formed, e.g.
Radicals:
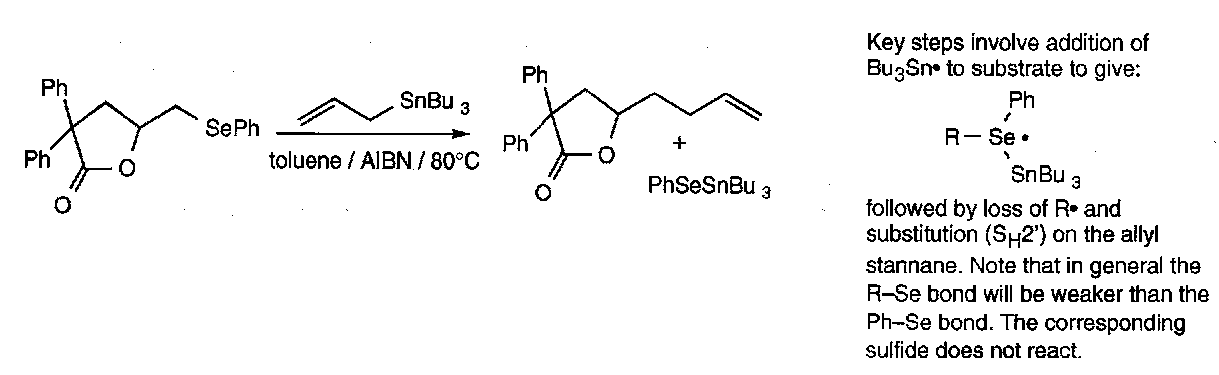
Radicals can attack selenides leading to hypervalent intermediates that subsequently fragments with cleavage of the weaker C-Se bond.
Generation of radicals in this manner is a particularly valuable route to acyl radicals from acyl aryl selenides.
Also,
Alkene Formation:
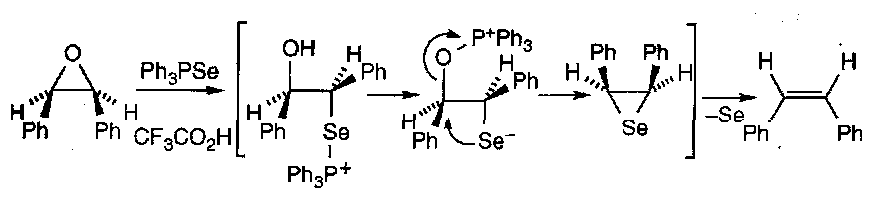
The weak C-Se bond allows alkenes to be formed from episelenides, with loss of selenium:
Note that this allows hindered alkenes to be obtained directly from selones and diazoalkanes.
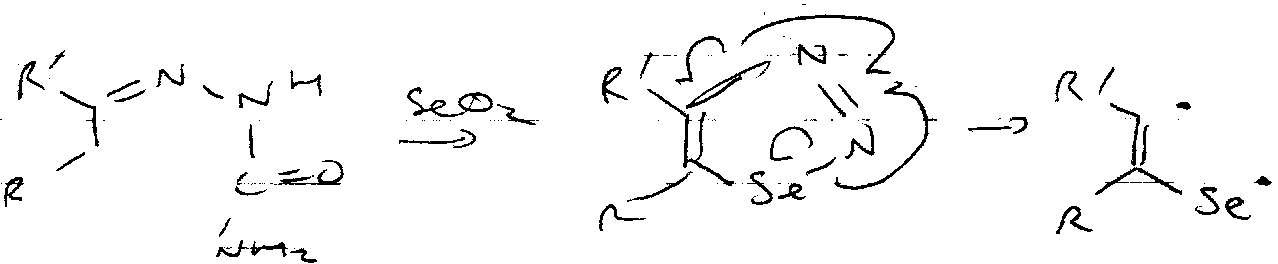
Eliminations:
Very similar to that for PhSSPh mechanism (i.e. includes a pericyclic sigmatropic shift). Also works with PhSeSePh (weak Se-Se bond, and SePh leaving groups).
Advantages are that it is SYN stereospecific and non-acidic.
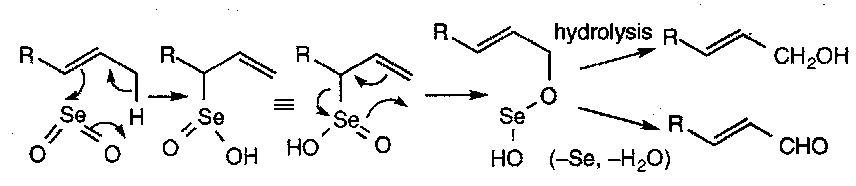
The Ene Reaction:
Note that the Se(OH)2 product can be re-oxidised (e.g. by a peroxide), which allows catalytic amounts of SeO2 to be used.
Also worth noting that instead of hydrolysing at the final step, Se and H2O can be removed to give the conjugated ketone.
PALLADIUM
The Heck Reaction
Note how the Heck coupling proceed at a terminal alkene:
The Suzuki Reaction
Thallium hydroxide can dramatically increase the rate of this coupling.
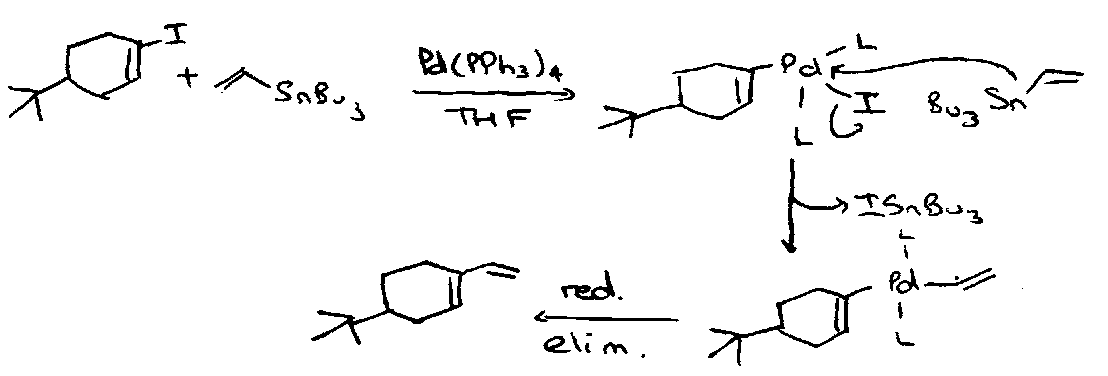
The Stille Reaction
I am happy for them to be reproduced, but please include credit to this website if you're putting them somewhere public please!