Alicyclic Chemistry
Basic notes on conformation and reactivity.
Alicyclic Chemistry Notes
More complicated due to the many available conformations. Different conformations may show different stereoselectivity. Need conformational bias to allow existing stereocentres to allow the creation of new ones (i.e. asymmetric induction).
Conformation
Cyclopropane, cyclobutane, cyclopentane – almost planar (dependent on substituents).
Cyclopropane has eclipsing CH2 units, and sp2 character at the carbons.
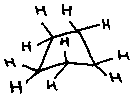
Cyclobutane puckers –
Non-planar geometry reduces torsional strain (Baeyer strain). Equatorial Br’s are more stable.
Cyclopentane –
Envelope conformation favoured to minimise eclipsing interactions (Pfitzer Strain).
Cyclohexane –
Three forms, of which chair is the most stable (eclipsing).
Twisted boat is flexible, so there is a large positive entropy gain. The energy profile looks like this:
Decalins –
The trans decalin is fixed, with a “flat” C skeleton an ax,ax bridge substituents:
The cis decalin is a flexible, curved molecule with ax,eq bridge substituents.
Energetics
Angle (Baeyer) Strain – due to angles not being 109o, i.e. when 3/4 membered rings, or when double bonds present. Both lead to decrease in angle. 8-10 membered rings increase bond angle.
Torsional (Pitzer) Strain – can’t rotate, so functional groups interact. Hence, not planar (less eclipsing).
This interaction is very significant in determining mechanisms and syntheses. For example,
Also, for similar reasons, a tertiary butyl group or similar alkyl is always equatorially placed, to avoid these bad interactions.
Increase in ring size increases negative entropy of Activation for the reaction. Thus, forming C = 3/4 is irreversible and favoured thermodynamically, but the activation energy is often too high due to Baeyer Strain. However, rings are typically 6 membered due to kinetic factors (and prevents larger ring formation).
Stereoselectivity
Baldwin’s Rules
Guidelines for predicting the relative facility of ring-forming reactions.
Ring closure reactions can be characterised in terms of three parameters:
5 - exo - trig
(a) (b) (c)
a) represents the number of atoms in the ring being formed.
b) indicates that the breaking bond ends up outside the final ring – exocyclic, cf. endo:
c) denotes the geometry of the atom being attacked (*) – trigonal (sp2) in this case, cf. tet and dig.
The following rules can then be applied to suggest the favoured or disfavoured nature of ring closure, based on the stereoelectronic requirements of the transition state:
|
Ring Size |
Exo |
Endo |
||||
|
tet (sp3) |
trig (sp2) |
dig (sp) |
tet (sp3) |
trig (sp2) |
dig (sp) |
|
|
3 |
✓ |
✓ |
✗ |
|
✗ |
✓ |
|
4 |
✓ |
✓ |
✗ |
|
✗ |
✓ |
|
5 |
✓ |
✓ |
✓ |
✗ |
✗ |
✓ |
|
6 |
✓ |
✓ |
✓ |
✗ |
✓ |
✓ |
|
7 |
✓ |
✓ |
✓ |
|
✓ |
✓ |
So 5-endo-trig is disfavoured:
The system is flat and little overlap is possible between the nitrogen lone pair and the LUMO (π*) of the sp2 carbon being attacked.
While 5-exo-trig is favoured:
Good overlap between the nitrogen lone pair and π* orbital of the carbonyl group is possible.
The Anomeric Effect
(more on this in Stereoelectronics Notes)
Reactivity
Steric environment effects according to formation of a transition state which involves an increase or decrease in bulk at the site of reaction.
Maintaining sterically desirable arrangements, e.g. antiperiplanar for base elimination.
Hydrolysis –
This is faster when equatorial, i.e. less compressed site.
Acetolysis of OTs:
Decrease in compression in Transition State due to trigonal arrangement. Faster when axial. This is due to:
Decrease in bulk at rate determining step, so faster when axial.
Cyclopropanes
The classic method of synthesis for these is the Simmons-Smith Reaction:
Another method involves the formation of a carbene (see Reactive Intermediates Tutorial):
Cyclobutanes
Heating these leads to the formation of ethene, while photochemical excitation of ethene molecules gives the cyclobutane.
Can also form from intramolecular Claisen Condensation (see cyclopentanes).
Cyclopentanes
These are kinetically much more favourable to form than the smaller rings.
Typical formation reactions are the Dieckmann Condensation (an intramolecular Claisen Condensation):
Also the Acyloin Condensation:
The Thorpe Reaction is also useful.
These can all be extended to six-membered rings by using a longer starting material. The Thorpe Reaction is also applicable to even larger rings.
Other Alicyclic Reactions
Elimination – must be antiperiplanar.
Transannular Reaction –
Occurs for C = 8-11.
Cycloaddition –
This includes the Diels-Alder Reactions:
(More on these in the Pericyclics Tutorial).
Demjanov –
Contraction –
Favorskii –
Robinson Ring Annelation –
Reactions with Decalins –
I am happy for them to be reproduced, but please include credit to this website if you're putting them somewhere public please!